CELL MEMBRANE
The structure and function of cells are critically dependent on membranes, which not only separate the interior of the cell from its environment but also define the internal compartments of eukaryotic cells, including the nucleus and cytoplasmic organelles. The formation of biological membranes is based on the properties of lipids, and all cell membranes share a common structural organization: bilayers of phospholipids with associated proteins. These membrane proteins are responsible for many specialized functions; some act as receptors that allow the cell to respond to external signals, some are responsible for the selective transport of molecules across the membrane, and others participate in electron transport and oxidative phosphorylation. In addition, membrane proteins control the interactions between cells of multicellular organisms. The common structural organization of membranes thus underlies a variety of biological processes and specialized membrane functions.
MEMBRANE COMPOSITION:
Plasma membrane is formed of the following chemical components. In addition to being composed of different components, the relative amounts of these components can vary from membrane to membrane. The table below shows some of the variation in protein, lipid and carbohydrate content of some membranes.
|
membrane |
% protein |
% lipid |
% carbohydrate |
|
myelin |
18 |
79 |
3 |
|
human erythrocyte plasma membrane |
49 |
43 |
8 |
|
mitochondrial inner membrane |
79 |
24 |
0 |
|
amoeba plasma membrane |
54 |
42 |
4 |
Membrane Lipids:
The fundamental building blocks of all cell membranes are phospholipids, which are amphipathic molecules, consisting of two hydrophobic fatty acid chains linked to a phosphate-containing hydrophilic head group. Because their fatty acid tails are poorly soluble in water, phospholipids spontaneously form bilayers in aqueous solutions, with the hydrophobic tails buried in the interior of the membrane and the polar head groups exposed on both sides, in contact with water. Such phospholipid bilayers form a stable barrier between two aqueous compartments and represent the basic structure of all biological membranes.
Lipids constitute approximately 50% of the mass of most cell membranes, although this proportion varies depending on the type of membrane. Plasma membranes, for example, are approximately 50% lipid and 50% protein. The inner membrane of mitochondria, on the other hand, contains an unusually high fraction (about 75%) of protein, reflecting the abundance of protein complexes involved in electron transport and oxidative phosphorylation. The lipid composition of different cell membranes also varies (Table 2.1). The plasma membrane of E. coli consists predominantly of phosphatidylethanolamine, which constitutes 80% of total lipid. Mammalian plasma membranes are more complex, containing four major phospholipids—phosphatidylcholine, phosphatidylserine, phosphatidylethanolamine, and sphingomyelin—which together constitute 50 to 60% of total membrane lipid. In addition to the phospholipids, the plasma membranes of animal cells contain glycolipids and cholesterol, which generally correspond to about 40% of the total lipid molecules.
|
Lipid Composition of Cell Membranes a |
||||
|
Lipid |
Plasma membrane |
Rough endoplasmic reticulum |
Outer mitochondrial membranes |
|
|
E. coli |
Erthrocyte |
|||
|
Phosphatidylcholine |
0 |
17 |
55 |
50 |
|
Phosphatidylserine |
0 |
6 |
3 |
2 |
|
Phosphatidylethanolamine |
80 |
16 |
16 |
2 |
|
Sphingomyelin |
0 |
17 |
3 |
5 |
|
Glycolipids |
0 |
2 |
0 |
0 |
|
Cholesterol |
0 |
45 |
6 |
<5 |
|
a Membrane compositions are indicated as the mole percentages of major lipid constituents. |
||||
Phospholipids:


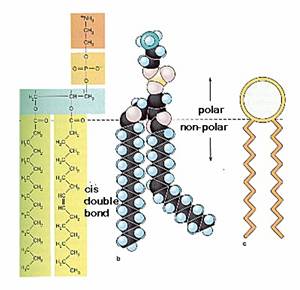
An important property of lipid bilayers is that they behave as two-dimensional fluids in which individual molecules (both lipids and proteins) are free to rotate and move in lateral directions (Fig 2.1). Such fluidity is a critical property of membranes and is determined by both temperature and lipid composition. For example, the interactions between shorter fatty acid chains are weaker than those between longer chains, so membranes containing shorter fatty acid chains are less rigid and remain fluid at lower temperatures. Lipids containing unsaturated fatty acids similarly increase membrane fluidity because the presence of double bonds introduces kinks in the fatty acid chains, making them more difficult to pack together.
The plasma membrane is the most thoroughly studied of all cell membranes, and it is largely through investigations of the plasma membrane that our current concepts of membrane structure have evolved. The plasma membranes of mammalian red blood cells (erythrocytes) have been particularly useful as a model for studies of membrane structure. Mammalian red blood cells do not contain nuclei or internal membranes, so they represent a source from which pure plasma membranes can be easily isolated for biochemical analysis. Indeed, studies of the red blood cell plasma membrane provided the first evidence that biological membranes consist of lipid bilayers. In 1925, two Dutch scientists (E. Gorter and R. Grendel) extracted the membrane lipids from a known number of red blood cells, corresponding to a known surface area of plasma membrane. They then determined the surface area occupied by a monolayer of the extracted lipid spread out at an air-water interface. The surface area of the lipid monolayer turned out to be twice that occupied by the erythrocyte plasma membranes, leading to the conclusion that the membranes consisted of lipid bilayers rather than monolayers.
The plasma membranes of animal cells contain four major phospholipids (phosphatidylcholine, phosphatidylethanolamine, phosphatidylserine, and sphingomyelin), which together account for more than half of the lipid in most membranes. These phospholipids are asymmetrically distributed between the two halves of the membrane bilayer. The outer leaflet of the plasma membrane consists mainly of phosphatidylcholine and sphingomyelin, whereas phosphatidylethanolamine and phosphatidylserine are the predominant phospholipids of the inner leaflet. A fifth phospholipid, phosphatidylinositol, is also localized to the inner half of the plasma membrane. Although phosphatidylinositol is a quantitatively minor membrane component, it plays an important role in cell signaling, as discussed in the next chapter. The head groups of both phosphatidylserine and phosphatidylinositol are negatively charged, so their predominance in the inner leaflet results in a net negative charge on the cytosolic face of the plasma membrane.
In addition to the phospholipids, the plasma membranes of animal cells contain glycolipids and cholesterol. The glycolipids are found exclusively in the outer leaflet of the plasma membrane, with their carbohydrate portions exposed on the cell surface. They are relatively minor membrane components, constituting only about 2% of the lipids of most plasma membranes. Cholesterol, on the other hand, is a major membrane constituent of animal cells, being present in about the same molar amounts as the phospholipids.
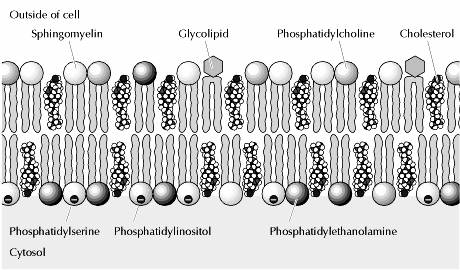
Another type of lipid in the membrane is cholesterol. The amount of cholesterol may vary with the type of membrane. Plasma membranes have nearly one cholesterol residue per phospholipid molecule. Other membranes (like those around bacteria) have no cholesterol. The cholesterol molecule inserts itself in the membrane with the same orientation as the phospholipid molecules. The figures show phospholipid molecules with a cholesterol molecule inbetween. Note that the polar head of the cholesterol is aligned with the polar head of the phospholipids.
Cholesterol molecules have several functions in the membrane:
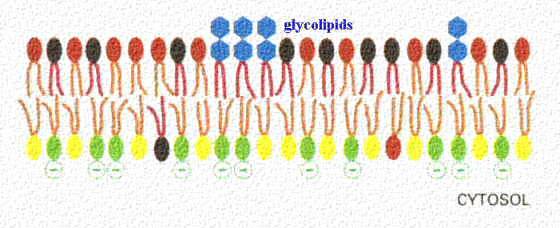
Glycolipids are also a constituent of membranes. In this figure, they are shown as blue sugar groups projecting into the extracellular space. They may microaggregate in the membrane. These components of the membrane may be protective, insulators, and sites of receptor binding. Among the molecules bound by glycososphingolipids include cell poisons such as cholera and tetanus toxins. The lower figure shows the chemical structure of two examples of glycososphingolipids.
Sphingolipids and cholesterol work together to help cluster proteins in a region called a "microdomain". They function as "rafts" or platforms for the attachment of proteins as membranes are moved around the cell and also during signal transduction.
Two general features of phospholipid bilayers are critical to membrane function. First, the structure of phospholipids is responsible for the basic function of membranes as barriers between two aqueous compartments. Because the interior of the phospholipid bilayer is occupied by hydrophobic fatty acid chains, the membrane is impermeable to water-soluble molecules, including ions and most biological molecules. Second, bilayers of the naturally occurring phospholipids are viscous fluids, not solids. The fatty acids of most natural phospholipids have one or more double bonds, which introduce kinks into the hydrocarbon chains and make them difficult to pack together. The long hydrocarbon chains of the fatty acids therefore move freely in the interior of the membrane, so the membrane itself is soft and flexible. In addition, both phospholipids and proteins are free to diffuse laterally within the membrane—a property that is critical for many membrane functions.
Because of its rigid ring structure, cholesterol plays a distinct role in membrane structure. Cholesterol will not form a membrane by itself, but inserts into a bilayer of phospholipids with its polar hydroxyl group close to the phospholipid head groups. Depending on the temperature, cholesterol has distinct effects on membrane fluidity. At high temperatures, cholesterol interferes with the movement of the phospholipid fatty acid chains, making the outer part of the membrane less fluid and reducing its permeability to small molecules. At low temperatures, however, cholesterol has the opposite effect: By interfering with interactions between fatty acid chains, cholesterol prevents membranes from freezing and maintains membrane fluidity. Although cholesterol is not present in bacteria, it is an essential component of animal cell plasma membranes. Plant cells also lack cholesterol, but they contain related compounds (sterols) that fulfill a similar function.
Recent studies suggest that not all lipids diffuse freely in the plasma membrane. Instead, discrete membrane domains appear to be enriched in cholesterol and the sphingolipids (sphingomyelin and glycolipids). These clusters of sphingolipids and cholesterol are thought to form “rafts” that move laterally within the plasma membrane and may associate with specific membrane proteins. Although the functions of lipid rafts remain to be understood, they may play important roles in processes such as cell signaling and the uptake of extracellular molecules by endocytosis.
Membrane Proteins:
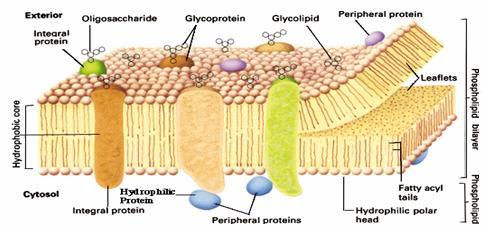
Proteins are the other major constituent of cell membranes, constituting 25 to 75% of the mass of the various membranes of the cell. The current model of membrane structure, proposed by Jonathan Singer and Garth Nicolson in 1972, views membranes as a fluid mosaic in which proteins are inserted into a lipid bilayer. While phospholipids provide the basic structural organization of membranes, membrane proteins carry out the specific functions of the different membranes of the cell.
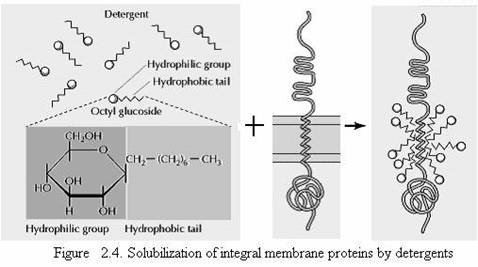
Singer and Nicolson distinguished two classes of membrane-associated proteins, which they called peripheral and integral membrane proteins. Peripheral membrane proteins were operationally defined as proteins that dissociate from the membrane following treatments with polar reagents, such as solutions of extreme pH or high salt concentration, that do not disrupt the phospholipid bilayer. Once dissociated from the membrane, peripheral membrane proteins are soluble in aqueous buffers. These proteins are not inserted into the hydrophobic interior of the lipid bilayer. Instead, they are indirectly associated with membranes through protein-protein interactions. These interactions frequently involve ionic bonds, which are disrupted by extreme pH or high salt. In contrast to the peripheral membrane proteins, integral membrane proteins can be released only by treatments that disrupt the phospholipid bilayer. Portions of these integral membrane proteins are inserted into the lipid bilayer, so they can be dissociated only by reagents that disrupt hydrophobic interactions. The most commonly used reagents for solubilization of integral membrane proteins are detergents, which are small amphipathic molecules containing both hydrophobic and hydrophilic groups (Fig 2.4). The hydrophobic portions of detergents displace the membrane lipids and bind to the hydrophobic portions of integral membrane proteins. Because the other end of the detergent molecule is hydrophilic, the detergent-protein complexes are soluble in aqueous solutions.
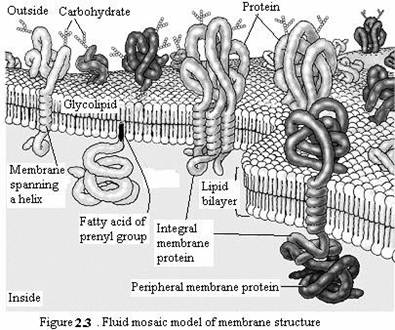
Many integral membrane proteins (called transmembrane proteins) span the lipid bilayer, with portions exposed on both sides of the membrane. These proteins can be visualized in electron micrographs of plasma membranes prepared by the freeze-fracture technique. In these specimens, the membrane is split and separates into its two leaflets. Transmembrane proteins are then apparent as particles on the internal faces of the membrane. The membrane-spanning portions of these proteins are usually α-helical regions of 20 to 25 nonpolar amino acids. The hydrophobic side chains of these amino acids interact with the fatty acid chains of membrane lipids, and the formation of an α helix neutralizes the polar character of the peptide bonds. Like the phospholipids, transmembrane proteins are amphipathic molecules, with their hydrophilic portions exposed to the aqueous environment on both sides of the membrane. Some transmembrane proteins span the membrane only once; others have multiple membrane-spanning regions. Most transmembrane proteins of eukaryotic plasma membranes have been modified by the addition of carbohydrates, which are exposed on the surface of the cell and may participate in cell-cell interactions. The membrane-spanning portions of transmembrane proteins are usually α helices of 20 to 25 hydrophobic amino acids that are inserted into the membrane of the endoplasmic reticulum during synthesis of the polypeptide chain. These proteins are then transported in membrane vesicles from the endoplasmic reticulum to the Golgi apparatus, and from there to the plasma membrane. Carbohydrate groups are added to the polypeptide chains in both the endoplasmic reticulum and Golgi apparatus, so most transmembrane proteins of the plasma membrane are glycoproteins with their oligosaccharides exposed on the surface of the cell.
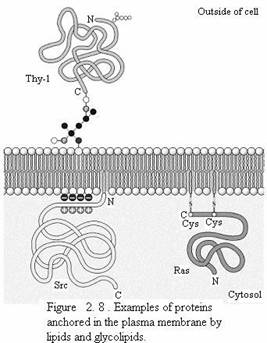
Proteins can also be anchored in membranes by lipids that are covalently attached to the polypeptide chain. Distinct lipid modifications anchor proteins to the cytosolic and extracellular faces of the plasma membrane. Proteins can be anchored to the cytosolic face of the membrane either by the addition of a 14-carbon fatty acid (myristic acid) to their amino terminus or by the addition of either a 16-carbon fatty acid (palmitic acid) or 15- or 20-carbon prenyl groups to the side chains of cysteine residues. Alternatively, proteins are anchored to the extracellular face of the plasma membrane by the addition of glycolipids to their carboxy terminus.
Studies of red blood cells have provided good examples of both peripheral and integral proteins associated with the plasma membrane. The membranes of human erythrocytes contain about a dozen major proteins, which were originally identified by gel electrophoresis of membrane preparations. Most of these are peripheral membrane proteins that have been identified as components of the cortical cytoskeleton, which underlies the plasma membrane and determines cell shape. For example, the most abundant peripheral membrane protein of red blood cells is spectrin, which is the major cytoskeletal protein of erythrocytes. Other peripheral membrane proteins of red blood cells include actin, ankyrin, and band 4.1. Ankyrin serves as the principal link between the plasma membrane and the cytoskeleton by binding to both spectrin and the integral membrane protein band 3. An additional link between the membrane and the cytoskeleton is provided by band 4.1, which binds to the junctions of spectrin and actin, as well as to glycophorin (the other major integral membrane protein of erythrocytes).
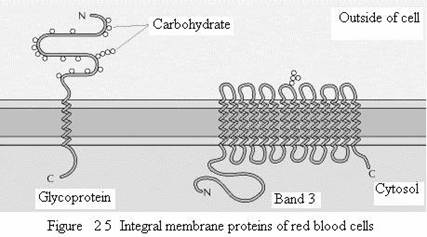
The two major integral membrane proteins of red blood cells, glycophorin and band 3, provide well-studied examples of transmembrane protein structure. Glycophorin is a small glycoprotein of 131 amino acids, with a molecular weight of about 30,000, half of which is protein and half carbohydrate. Glycophorin crosses the membrane with a single membrane-spanning α helix of 23 amino acids, with its glycosylated amino-terminal portion exposed on the cell surface. Although glycophorin was one of the first transmembrane proteins to be characterized, its precise function remains unknown. In contrast, the function of the other major transmembrane protein of red blood cells is well understood. This protein, originally known as band 3, is the anion transporter responsible for the passage of bicarbonate (HCO3-) and chloride (Cl-) ions across the red blood cell membrane. The band 3 polypeptide chain is 929 amino acids and is thought to have 14 membrane-spanning α-helical regions. Within the membrane, dimers of band 3 form globular structures containing internal channels through which ions are able to travel across the lipid bilayer.
Glycocalyx:
The extracellular portions of plasma membrane proteins are generally glycosylated. Likewise, the carbohydrate portions of glycolipids are exposed on the outer face of the plasma membrane. Consequently, the surface of the cell is covered by a carbohydrate coat, known as the glycocalyx, formed by the oligosaccharides of glycolipids and transmembrane glycoproteins.
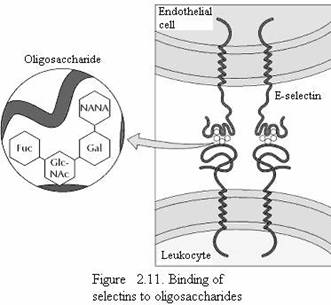
Part of the role of the glycocalyx is to protect the cell surface. In addition, the oligosaccharides of the glycocalyx serve as markers for a variety of cell-cell interactions. A well-studied example of these interactions is the adhesion of white blood cells (leukocytes) to the endothelial cells that line blood vessels—a process that allows the leukocytes to leave the circulatory system and mediate the inflammatory response in injured tissues. The initial step in adhesion between leukocytes and endothelial cells is mediated by a family of transmembrane proteins called selectins, which recognize specific carbohydrates on the cell surface (Fig 2.11). Two members of the selectin family (E-selectin and P-selectin), expressed by endothelial cells and platelets, bind to specific oligosaccharides expressed on the surface of leukocytes. A different selectin (L-selectin) is expressed by leukocytes and recognizes an oligosaccharide on the surface of endothelial cells. The oligosaccharides exposed on the cell surface thus provide a set of markers that help identify the distinct cell types of multicellular organisms.
Membrane carbohydrates:
They form the cell coat around the plasma membrane. Hexose, Hexosamine, fucose and sialic acid are the important carbohydrates found in the plasma membrane of erythrocytes. Plasma membranes of Amoeba proteus contain large amount of polysaccharides.
Membrane Nucleic acid:
The plasma membrane of Arabacia egg contains nucleic acids.
Membrane Salts and Water:
Salts are generally present in the cell membrane. Some of them are present in higher concentration. The cell membrane also contains water.
MEMBRANE MODELS:
Membrane contains proteins and lipids mainly. These components are arranged in a definite pattern. The following models are proposed to explain the structure of plasma membrane.
1. Bimolecular Leaflet Model
2. Lattice Model
3. Micellar Model
4. Trilaminar Model and
5. Fluid Mosaic Model
Bimolecular Leaflet Model:
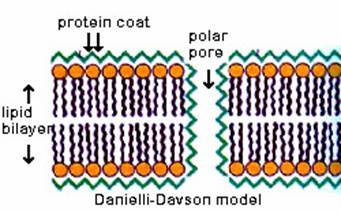
Danielli and Davson (1935) proposed a phospholipid interior with protein coats on both sides. Cell membrane is made of a phospholipid bilayer sandwiched between two layers of globular protein.
Though the phospholipid bilayer is probably accurate, there are problems with the Davson- Danielli model:
2. Lattice Model
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
This model was proposed by Wolpers in 1941. According to this model, lipids and proteins are not arranged in layers but as a lattice or network. Proteins form a kind of mechanical frame work. The lipid component is distributed between the meshes of the protein frame.
3. Micellar Model:
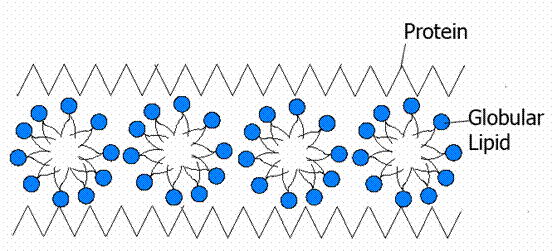
This model was proposed by Hilleir and Hoffman in 1953. According to this model, the molecules in the membrane are not arranged in layers; but they are arranged in the form of globular subunits called micelles. The lipid micelles are the building blocks of the membrane and the protein globules are arranged on either side.
4. Trilaminar Model (Unit membrane Model):
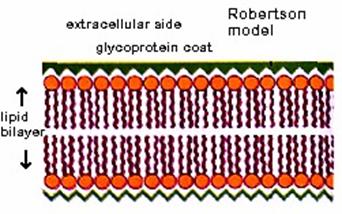
This model was proposed by Robertson in 1959. According to this model, the plasma membrane is formed of three layers. The three layers are an outer protein layer, a middle lipid layer and an inner protein layer. The middle layer is 235A thick and the inner and outer layers are 20A each. Such a trilaminar membrane is also called as unit membrane. Robertson believed that all biological membranes are unit membranes. The plasma membranes of prokaryotes and eukaryotes are unit membranes. Again the membranes of endoplasmic reticulum, Golgi bodies, Mitochondria, lysosomes, plastids and nucleus are unit membranes.
5. Fluid Mosaic Model:
In the year 1972 Singer and Nicolson proposed a model for explaining the membrane structure, taking into account all the known facts. According to this model, cell membrane consists of a highly viscous fluid matrix of a bilayer of phospholipids having globular proteins associated with them. This model came to be known as fluid mosaic model. The phospholipid molecules in the cell membrane have their polar, hydrophilic heads towards outer surface and the nonpolar, hydrophobic tails towards inner surface. This arrangement forms a water resistant barrier through which only lipid soluble substances can pass through. The proteins in cell membrane are of two kinds. Some of the proteins are found at the periphery, partly projecting out of the lipid layer. These are the extrinsic proteins. These proteins can be easily extracted. Some of the protein molecules are found totally embedded in the phospholipid matrix. Those are the intrinsic or integral proteins, which represent nearly 70% of the membrane proteins. These proteins cannot be extracted. Some of the integral proteins have very large molecules that extend throughout the phospholipid matrix, projecting out on both surfaces. These proteins are called tunnel proteins or transmembrane proteins. They are believed to have channels for the passage of water-soluble substances. The scattered arrangement of protein molecules in the phospholipid matrix gives the appearance of a mosaic pattern. Hence, the name to the model. A few molecules of oligosaccharides are found attached to the free ends of protein molecules on the outer surface of the membrane. Sometimes they are also found attached to phospholipid layer.
The cell membrane and the membranes of various cell structures exhibit the same structure and as such perform the same functions. However, the amount and arrangement of phospholipids and proteins varies in different cells and organelles. This variation accords a specificity that is unique to each membrane.
The bio-membranes exhibit the following properties
These properties indicate that biomembranes are fluid and dynamic.
SPECIALIZATION OF PLASMA MEMBRANE:
The plasma membrane shows some specialized structures. These may be due to outgrowths or ingrowths or contact with adjacent membrane. Such structures include the following:
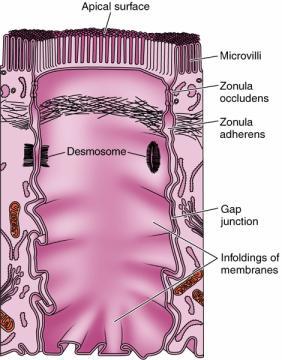
1. Microvilli:
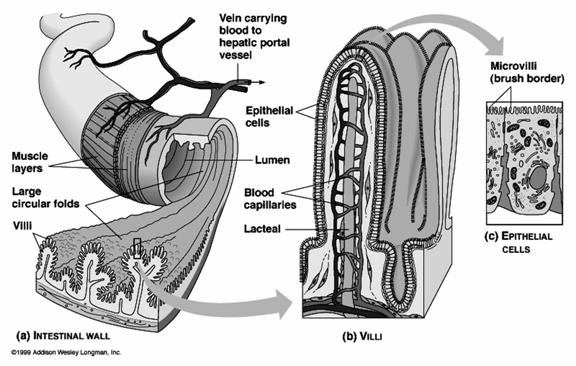
Microvilli are minute finger-like projections arising from the surface of certain cells. They are found on the epithelial cells of intestine, kidney tubules, gall bladder, uterus, hepatic cells and yolk cells. A single cell contains about 3000 microvilli. Each microvillus is cylindrical in shape. The microvillus is 0.6 -0.8 micrometer long and has a diameter of 0.1micrometer. It has a core of cytoplasm enclosed by the plasma membrane. The cytoplasmic core is transversed by the fine microfilaments made up of actin. The micro filaments are attached to the tip of of the microvillus by a-actin. The micro filaments give rigidity to the microvilli. The microvilli increase the surface area of the cell and help in effective absorption. The narrow spaces lying between the microvilli from a kind of sieve through which substances pass during the process of absorption.
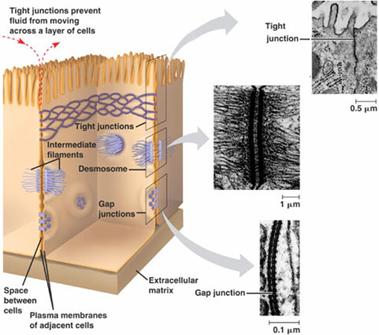
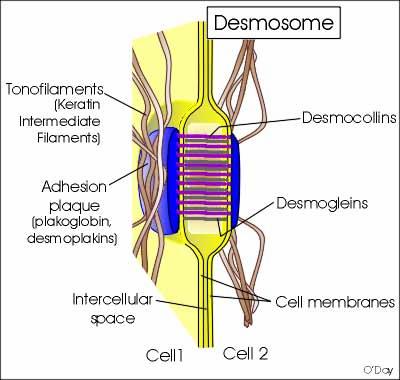
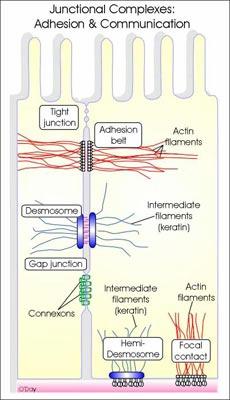
2. Desmosomes:
Desmosomes are thickened areas of plasma membranes of two adjacent cells from which radiate fine tonofibrils and which provide adhesion to the cell. Desmosomes are abundant in cardiac muscles and skin that are subject to sever mechanical stress. The tonofibrils provide intracellular mechanical support. In the desmosome, the intracellular gap is filled with a dense fibrous material of acid mucosaccharides and proteins. This material helps to glue the cells together.
There are four types of desmosomes namely belt, spot, hemi and septate desmosomes.
Belt desmosome – It is found just below the tight junction. It is present as a band on the inner side of the cell membrane of the cells.
Spot desmosome – It is disc shaped points of contact between the plasma membrane of adjacent cells.
Hemi desmosome- It is half desmosome. It is present at the basal surface of the certain epithelial cells.
Septate desmosome – It is a desmosome containing many transverse septa between the plasma membrane in the intracellular space.
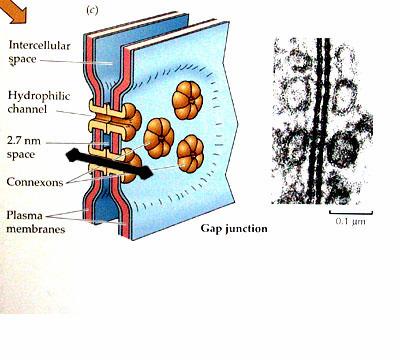
3. Gap Junctions:
Gap junctions, which are found in most animal tissues, serve as direct connections between the cytoplasms of adjacent cells(Fig 2.56). They provide open channels through the plasma membrane, allowing ions and small molecules (less than approximately a thousand daltons) to diffuse freely between neighboring cells, but preventing the passage of proteins and nucleic acids. Consequently, gap junctions couple both the metabolic activities and the electric responses of the cells they connect. Most cells in animal tissues—including epithelial cells, endothelial cells, and the cells of cardiac and smooth muscle—communicate by gap junctions. In electrically excitable cells, such as heart muscle cells, the direct passage of ions through gap junctions couples and synchronizes the contractions of neighboring cells. Gap junctions also allow the passage of some intracellular signaling molecules, such as cAMP and Ca2+, between adjacent cells, potentially coordinating the responses of cells in tissues.
4. Tight Junctions:
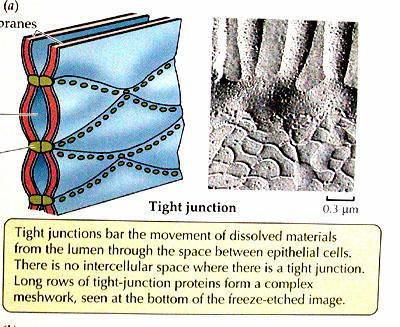
Tight junctions are the closest known contacts between adjacent cells. They were originally described as sites of apparent fusion between the outer leaflets of the plasma membranes, although it is now clear that the membranes do not fuse. Instead, tight junctions appear to be formed by a network of protein strands that continues around the entire circumference of the cell. Each strand in these networks is thought to be composed of transmembrane proteins (claudins and occludin) that bind to similar proteins on adjacent cells, thereby sealing the space between their plasma membranes.
5. Inter-digitations:
The plasma membranes of adjacent cells project into the cytoplasm as finger-like projections called interdigitations. They help to compartmentalize the cytoplasm.
6. Balsal infoldings:
Basal infoldings are finger-like invaginations of the plasma membrane into the cytoplasm from the base of the cell. They are found in kidney cells. The basal infoldings function as septa and they split the cytoplasm into compartments. They enclose many mitochondria. They are concerned with the active transport of materials.
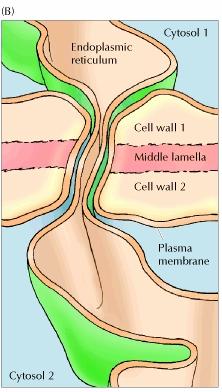
7. Plasmodesmata:
Adhesion between plant cells is mediated by their cell walls rather than by transmembrane proteins. In particular, a specialized pectin-rich region of the cell wall called the middle lamella acts as a glue to hold adjacent cells together. Because of the rigidity of plant cell walls, stable associations between plant cells do not require the formation of cytoskeletal links, such as those provided by the desmosomes and adherens junctions of animal cells. However, adjacent plant cells communicate with each other through cytoplasmic connections called plasmodesmata (singular, plasmodesma), which function analogously to animal cell gap junctions.
Despite their similarities in function, plasmodesmata are structurally unrelated to gap junctions. At each plasmodesma the plasma membrane of one cell is continuous with that of its neighbor, creating an open channel between the two cytosols. An extension of the smooth endoplasmic reticulum passes through the pore, leaving a ring of surrounding cytoplasm through which ions and small molecules are able to pass freely between the cells. In addition, plasmodesmata can expand in response to appropriate stimuli, permitting the regulated passage of macromolecules between adjacent cells. Plasmodesmata may thus play a key role in plant development by controlling the trafficking of regulatory molecules, such as transcription factors or RNAs, between cells.
MEMBRANE FUNCTIONS:
The plasma membrane has the following functions:
1. Mechanical support:
Plasma membrane gives the shape to the cell. It protects cell contents and keeps the cell content in palce.
2. Exchange of material:
Plasma membrane regulates the exchange of materials into and out of the cell. It allows the needed materials to enter the cell and sends out the unwanted materials from the cell. Hence the cell membrane allows one substance to pass through more easily than another. This property of the cell membrane is said to be selective permeability.
3. Biogenesis of cell organelles:
Certain cell organelles like endoplasmic reticulum, nuclear membrane etc., develop from plasma membrane.
4. Absorption:
The microvilli of intestinal cells increase the surface area. Hence the rate of absorption increases.
5. Cell recognition:
Mammalian leucocytes recognize foreign cells like bacteria and engulf them by phagocytosis. Similarly the macrophages of spleen can identify worn out erythrocytes from healty cells and destroy them. The sites of cell recognition are located on the surface of the plasma membrane. The amino sugar sialic acid is involved in cell recognition.
6. Antigenic specificity:
The antigen specificities of the cells are located on the surface of the plasma membrane. The antigenic determinants are the glycoproteins of plasma membrane. The graft rejection of transplanted tissues is determined by antigens (glycoproteins) located on the cell membrane of implanted cells.
7. Transmission of impulses:
The plasma membrane of nerve fibres transmit nerve impulses during nerve impulse transmission.
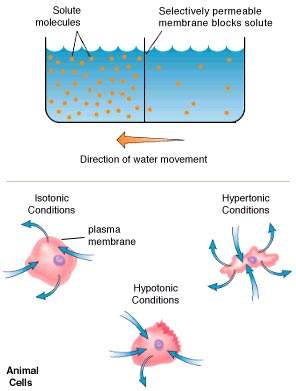
8. Osmosis:
The plasma membrane allows the free movement of water. The process of movement of water molecules from the region of higher water concerntation to the region of lower water concentration is known as osmosis. The process in which the water molecules enter the cell is known as endosmosis and the reverse processs is known as exo-osmosis. Due to endosmosis the pressure inside the cell increases. This pressure is termed as hydrostatic pressure. Since this pressure is caused by the osmosis, it is also called as osmotic pressure. The plasma membrane maintains a balance between the osmotic pressure of the inter and intra cellular fluids.
9. Endocytosis: (Refer lysosome notes)
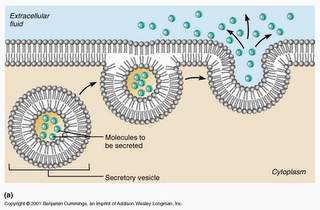
10. Exocytosis:
It is otherwise called as cell vomiting. The process of exudating the secretory products from the secretory cells to the outside of the cell cytoplasm are known as exocytosis. This process is also called emeiocytosis or reverse endocytosis. In pancreatice cells, the enzymatic secretions are passed out with the help of the plasma membrane.
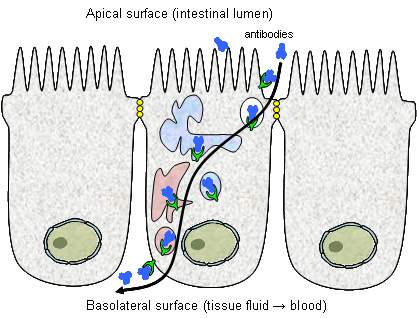
11. Transcytosis:
It is the transport of materials through a cell. It is also otherwise called as cytopemphis. The material passes into the cell by endocytosis and then it comes out of the cell by exocytosis without any change. Glucose molecules from the intestine lumen move into the blood capillary through the epithelial cells by transcytosis. Antibodies from intestinal lumen also transferred to blood through transcytosis.
12. Membrane Transport:
The selective permeability of biological membranes to small molecules allows the cell to control and maintain its internal composition. Only small uncharged molecules can diffuse freely through phospholipid bilayers. Small nonpolar molecules, such as O2 and CO2, are soluble in the lipid bilayer and therefore can readily cross cell membranes. Small uncharged polar molecules, such as H2O, also can diffuse through membranes, but larger uncharged polar molecules, such as glucose, cannot. Charged molecules, such as ions, are unable to diffuse through a phospholipid bilayer regardless of size; even H+ ions cannot cross a lipid bilayer by free diffusion. Gases, hydrophobic molecules, and small polar uncharged molecules can diffuse through phospholipid bilayers.
Although ions and most polar molecules cannot diffuse across a lipid bilayer, many such molecules (such as glucose) are able to cross cell membranes. These molecules pass across membranes via the action of specific transmembrane proteins, which act as transporters. Such transport proteins determine the selective permeability of cell membranes and thus play a critical role in membrane function. They contain multiple membrane-spanning regions that form a passage through the lipid bilayer, allowing polar or charged molecules to cross the membrane through a protein pore without interacting with the hydrophobic fatty acid chains of the membrane phospholipids.
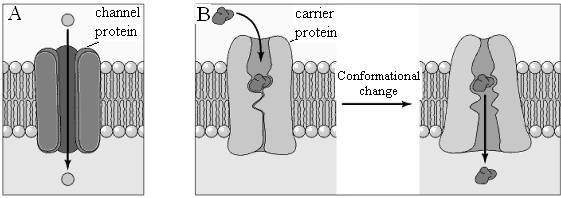
There are two general classes of membrane transport proteins. Channel proteins form open pores through the membrane, allowing the free passage of any molecule of the appropriate size. Ion channels, for example, allow the passage of inorganic ions such as Na+, K+, Ca2+, and Cl- across the plasma membrane. Once open, channel proteins form small pores through which ions of the appropriate size and charge can cross the membrane by free diffusion. The pores formed by these channel proteins are not permanently open; rather, they can be selectively opened and closed in response to extracellular signals, allowing the cell to control the movement of ions across the membrane. Such regulated ion channels have been particularly well studied in nerve and muscle cells, where they mediate the transmission of electrochemical signals.
In contrast to channel proteins, carrier proteins selectively bind and transport specific small molecules, such as glucose. Rather than forming open channels, carrier proteins act like enzymes to facilitate the passage of specific molecules across membranes. In particular, carrier proteins bind specific molecules and then undergo conformational changes that open channels through which the molecule to be transported can pass across the membrane and be released on the other side.
Molecules transported by either channel or carrier proteins cross membranes in the energetically favorable direction, as determined by concentration and electrochemical gradients—a process known as passive transport. However, carrier proteins also provide a mechanism through which the energy changes associated with transporting molecules across a membrane can be coupled to the use or production of other forms of metabolic energy, just as enzymatic reactions can be coupled to the hydrolysis or synthesis of ATP. For example, molecules can be transported in an energetically unfavorable direction across a membrane (e.g., against a concentration gradient) if their transport in that direction is coupled to ATP hydrolysis as a source of energy—a process called active transport. The free energy stored as ATP can thus be used to control the internal composition of the cell, as well as to drive the biosynthesis of cell constituents.
Passive Transport:
It occurs in two ways namely Simple diffusion and Facilitated diffusion.
Simple diffusion:
The simplest mechanism by which molecules can cross the plasma membrane is simple diffusion. During simple passive diffusion, a molecule simply dissolves in the phospholipid bilayer, diffuses across it, and then dissolves in the aqueous solution at the other side of the membrane. No membrane proteins are involved and the direction of transport is determined simply by the relative concentrations of the molecule inside and outside of the cell. The net flow of molecules is always down their concentration gradient—from a compartment with a high concentration to one with a lower concentration of the molecule.
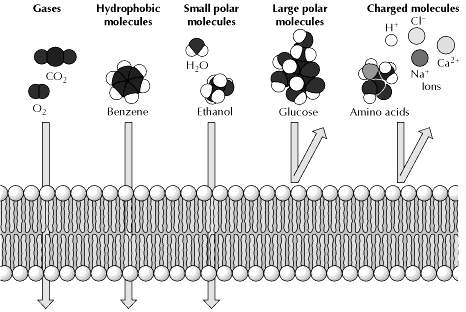
Simple passive diffusion is thus a nonselective process by which any molecule able to dissolve in the phospholipid bilayer is able to cross the plasma membrane and equilibrate between the inside and outside of the cell. Importantly, only small, relatively hydrophobic molecules are able to diffuse across a phospholipid bilayer at significant rates. Thus, gases (such as O2 and CO2), hydrophobic molecules (such as benzene), and small polar but uncharged molecules (such as H2O and ethanol) are able to diffuse across the plasma membrane.
Facilitated Diffusion:
Facilitated diffusion, like passive diffusion, involves the movement of molecules in the direction determined by their relative concentrations inside and outside of the cell. No external source of energy is provided, so molecules travel across the membrane in the direction determined by their concentration gradients and, in the case of charged molecules, by the electric potential across the membrane. However, facilitated diffusion differs from passive diffusion in that the transported molecules do not dissolve in the phospholipid bilayer. Instead, their passage is mediated by proteins that enable the transported molecules to cross the membrane without directly interacting with its hydrophobic interior. Facilitated diffusion therefore allows polar and charged molecules, such as carbohydrates, amino acids, nucleosides, and ions, to cross the plasma membrane.
Two classes of proteins that mediate facilitated diffusion are generally distinguished: carrier proteins and channel proteins. Carrier proteins bind specific molecules to be transported on one side of the membrane. They then undergo conformational changes that allow the molecule to pass through the membrane and be released on the other side. In contrast, channel proteins (see the next section) form open pores through the membrane, allowing the free diffusion of any molecule of the appropriate size and charge.
Carrier Proteins:
Carrier proteins are responsible for the facilitated diffusion of sugars, amino acids, and nucleosides across the plasma membranes of most cells. The uptake of glucose, which serves as a primary source of metabolic energy, is one of the most important transport functions of the plasma membrane, and the glucose transporter provides a well-studied example of a carrier protein. The glucose transporter was initially identified as a 55-kd protein in human red blood cells, in which it represents approximately 5% of total membrane protein. The binding of glucose to this exterior site induces a conformational change in the transporter, such that the glucose-binding site now faces the interior of the cell. Glucose can then be released into the cytosol, followed by the return of the transporter to its original conformation.
Most cells, including erythrocytes, are exposed to extracellular glucose concentrations that are higher than those inside the cell, so facilitated diffusion results in the net inward transport of glucose. Once glucose is taken up by these cells it is rapidly metabolized, so intracellular glucose concentrations remain low and glucose continues to be transported into the cell from the extracellular fluids. Because the conformational changes of the glucose transporter are reversible, however, glucose can be transported in the opposite direction simply by reversing the steps. Such reverse flow occurs, for example, in liver cells, in which glucose is synthesized and released into the circulation.
Channels:
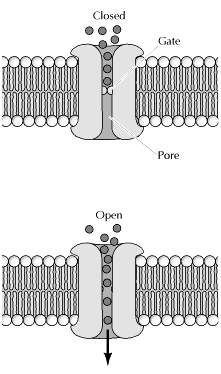
In contrast to carrier proteins, channel proteins simply form open pores in the membrane, allowing small molecules of the appropriate size and charge to pass freely through the lipid bilayer. One group of channel proteins, discussed earlier, is the porins, which permit the free passage of ions and small polar molecules through the outer membranes of bacteria. Channel proteins also permit the passage of molecules between cells connected at gap junctions, which are discussed later in the chapter. The plasma membranes of many cells also contain water channel proteins (aquaporins), through which water molecules are able to cross the membrane much more rapidly than they can diffuse through the phospholipid bilayer. The best-characterized channel proteins, however, are the ion channels, which mediate the passage of ions across plasma membranes. Although ion channels are present in the membranes of all cells, they have been especially well studied in nerve and muscle, where their regulated opening and closing is responsible for the transmission of electric signals.
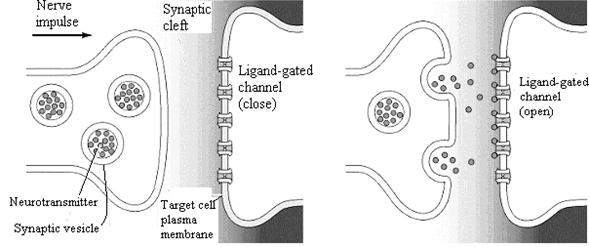
Three properties of ion channels are central to their function. First, transport through channels is extremely rapid. More than a million ions per second flow through open channels—a flow rate approximately a thousand times greater than the rate of transport by carrier proteins. Second, ion channels are highly selective because narrow pores in the channel restrict passage to ions of the appropriate size and charge. Thus, specific channel proteins allow the passage of Na+, K+, Ca2+, and Cl- across the membrane. Third, most ion channels are not permanently open. Instead, the opening of ion channels is regulated by “gates” that transiently open in response to specific stimuli. Some channels (called ligand-gated channels) open in response to the binding of neurotransmitters or other signaling molecules; others (voltage-gated channels) open in response to changes in electric potential across the plasma membrane.
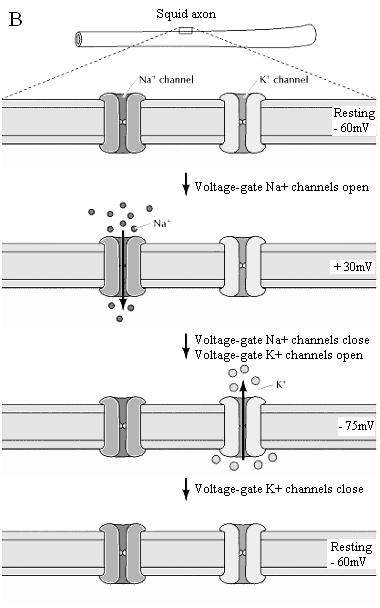
Active Transport:
The net flow of molecules by facilitated diffusion, through either carrier proteins or channel proteins, is always energetically downhill in the direction determined by electrochemical gradients across the membrane. In many cases, however, the cell must transport molecules against their concentration gradients. In active transport, energy provided by another coupled reaction (such as the hydrolysis of ATP) is used to drive the uphill transport of molecules in the energetically unfavorable direction.
Na+-K+ Pumb driven by ATP:
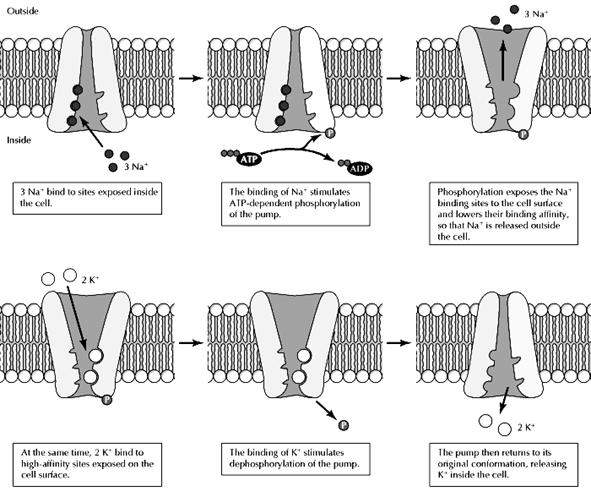
The ion pumps responsible for maintaining gradients of ions across the plasma membrane provide important examples of active transport driven directly by ATP hydrolysis.The concentration of Na+ is approximately ten times higher outside than inside of cells, whereas the concentration of K+ is higher inside than out. These ion gradients are maintained by the Na+-K+ pump (also called the Na+-K+ ATPase), which uses energy derived from ATP hydrolysis to transport Na+ and K+ against their electrochemical gradients. This process is a result of ATP-driven conformational changes in the pump. First, Na+ ions bind to high-affinity sites inside the cell. This binding stimulates the hydrolysis of ATP and phosphorylation of the pump, inducing a conformational change that exposes the Na+-binding sites to the outside of the cell and reduces their affinity for Na+. Consequently, the bound Na+ is released into the extracellular fluids. At the same time, high-affinity K+-binding sites are exposed on the cell surface. The binding of extracellular K+ to these sites then stimulates hydrolysis of the phosphate group bound to the pump, which induces a second conformational change, exposing the K+-binding sites to the cytosol and lowering their binding affinity so that K+ is released inside the cell. The pump has three binding sites for Na+ and two for K+, so each cycle transports three Na+ and two K+ across the plasma membrane at the expense of one molecule of ATP.
Active Transport driven by Ion gradients:
The Na+ gradient established by the Na+-K+ pump provides a source of energy that is frequently used to power the active transport of sugars, amino acids, and ions in mammalian cells. The H+ gradients established by the H+ pumps of bacteria, yeast, and plant cells play similar roles.
The epithelial cells lining the intestine provide a good example of active transport driven by the Na+ gradient. These cells use active-transport systems in the apical domains of their plasma membranes to take up dietary sugars and amino acids from the lumen of the intestine. The uptake of glucose, for example, is carried out by a transporter that coordinately transports two Na+ and one glucose into the cell. The flow of Na+ down its electrochemical gradient provides the energy required to take up dietary glucose and to accumulate high intracellular glucose concentrations. Glucose is then released into the underlying connective tissue (which contains blood capillaries) at the basolateral surface of the intestinal epithelium, where it is transported down its concentration gradient by facilitated diffusion (Fig 2.31). The uptake of glucose from the intestinal lumen and its release into the circulation thus provides a good example of the polarized function of epithelial cells, which results from the specific localization of active transport and facilitated diffusion carriers to the apical and basolateral domains of the plasma membrane, respectively.
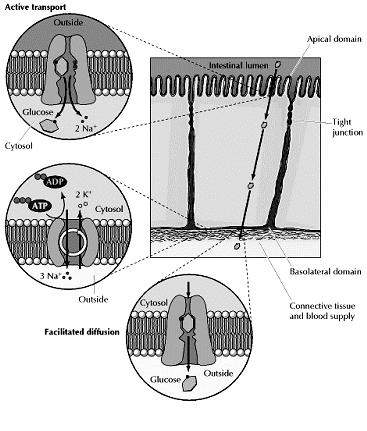
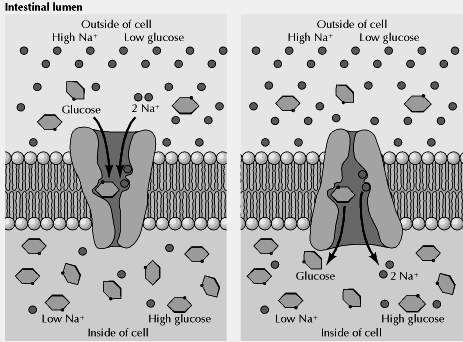
Co-transport:
Cotransport is the name of a process in which two substances are simultaneously transported across a membrane by one protein, or protein complex which does not have ATPase activity.
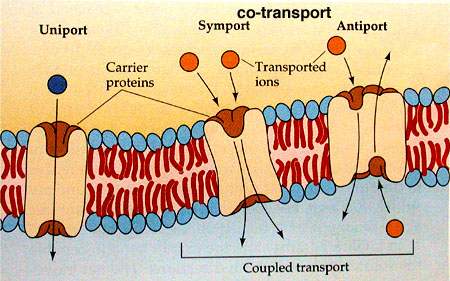
Different types of co-transport
Symport
When both substances are transported in the same direction the transport protein is known as a symport.
Antiport
When the substances are transported in opposite directions the transport protein is known as an antiport.