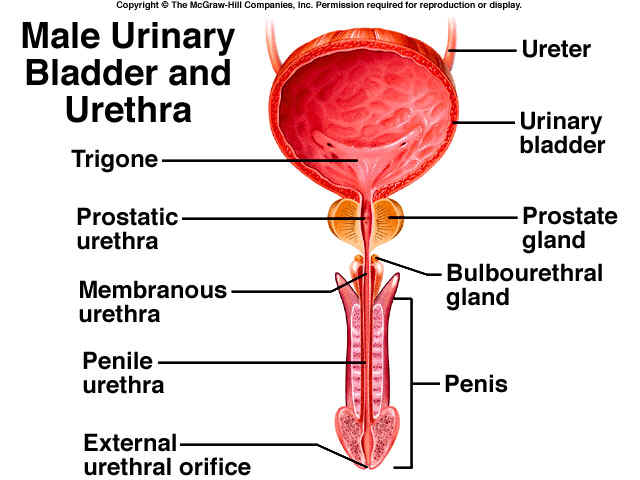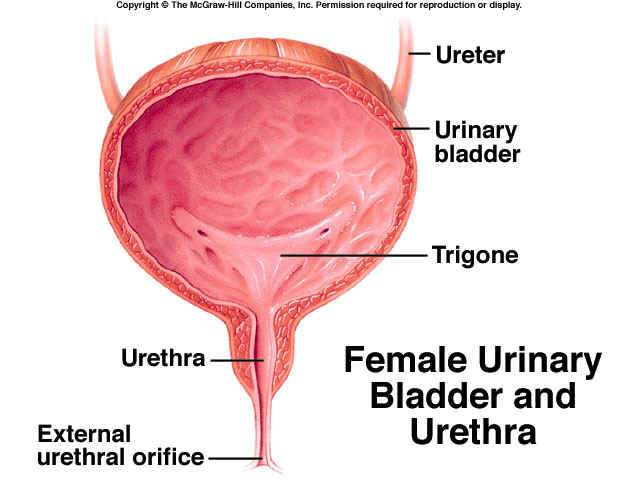
EXCRETION is the process by which substances, mostly waste products, are removed from the blood and eliminated from the body. Kidneys are the chief excretory organs that remove non-volatile fixed acids, apart from other substances. Thus, the kidneys should be emphasized to have “homeostatic” function, primarily.
FUNCTIONS OF THE KIDNEY
Ø Elimination of waste products of metabolism, especially, N2, S-containing products of protein metabolism.
Ø Water balance.
Ø Osmotic balance by maintaining ionic balance.
Ø Maintenance of optimal concentration of useful substances.
Ø Acid-base balance.
Ø Xenobiotics and drugs.
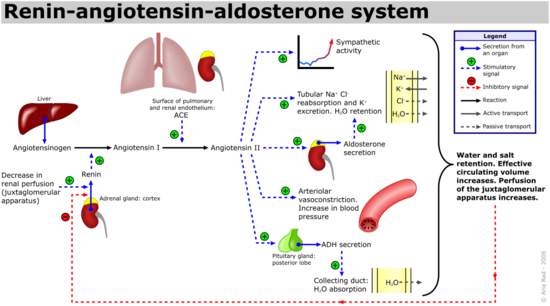
Ø Renin synthesis: The juxta-glomerular cells (JGC) release rennin, which induces AT-II synthesis which acts as a vasoconstrictor and aldosterone stimulator.
Ø Erythropoietin: The endothelial cells of peri-tubular capillaries synthesize this signal hormone for RBC synthesis.
Ø Conversion of 25-OH Vit-D to the active form 1, 25-(OH)2 Vit-D in PCT or pars recta.
Ø Prostaglandins: Synthesized by medullary interstitial type I cells. They cause ^ renin and also vaso-dilation and ^ Na excretion.
Ø Tissue kallikrien: It is a local hormone that cause vaso-dilation and ^ Na excretion.

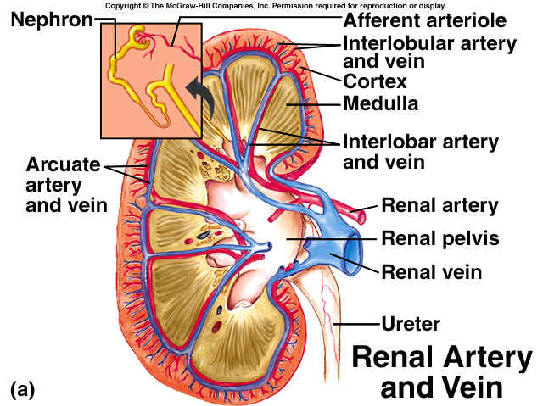
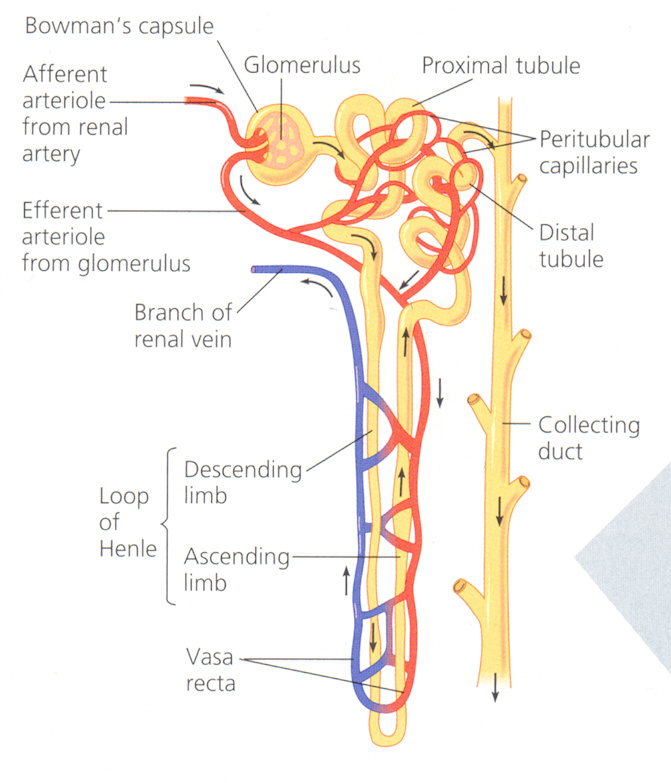
NEPHRON
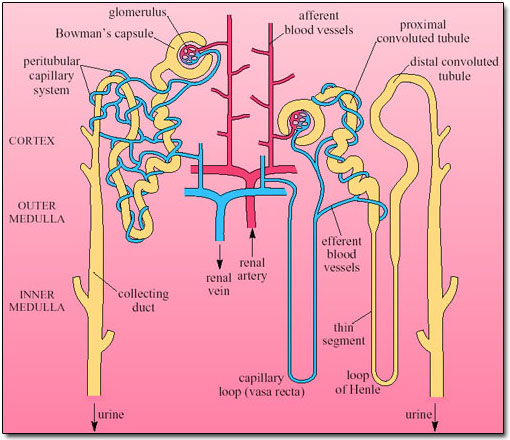
Nephron is the structural and functional unit of the kidneys. There are about 106 nephron per kidney. About 85% lie in the outer 2/3rd of the cortex and are called superficial or cortical nephrons. The remaining 15% are known as juxta-medullary nephrons that lie in the medulla. A nephron can be divided in to 2 major parts:
 BOWMANN’S
CAPSULE: It is a
blind dilatation of renal tubule having a crescentric cavity (0.2 mm).
BOWMANN’S
CAPSULE: It is a
blind dilatation of renal tubule having a crescentric cavity (0.2 mm).
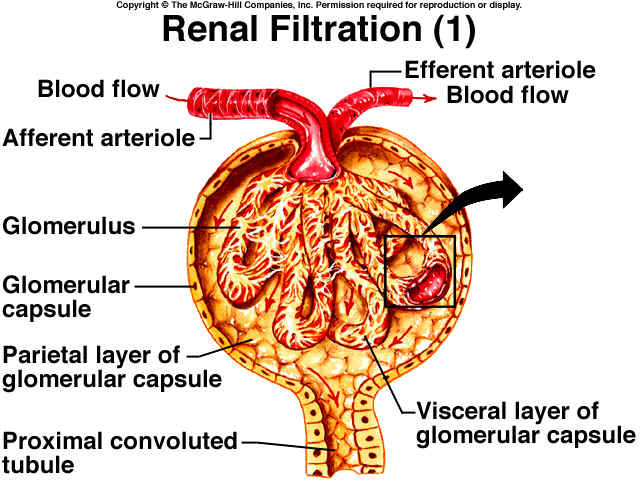
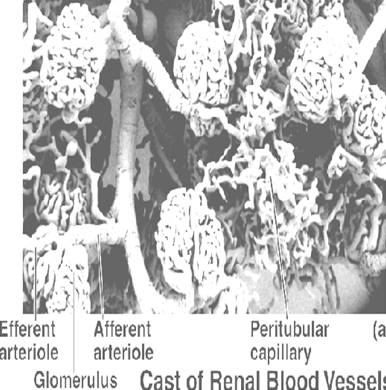
GLOMERULUS: It has about 50 capillaries formed by a tuft of afferent arterioles (50 µm diam.) arising from renal artery, and reunites to form efferent arterioles (25 µm).
FILTERING MEMBRANE: It is made of 5 layers, namely:
PROXIMAL CONVULUTED TUBULE: It contributes to about 1/3rd of the renal tubule is lined by a single layer of columnar epithelials in pars convuluta and cubical epithelials in pars recta. These cells have luminal brush borders or µ-villi that ^ ses the surface area for tubular reabsorption.
LOOP OF HENLE:
It is lined with squamous epithelials. It has 3 regions, a) thin descending
loop, b) ascending loop and c) thick ascending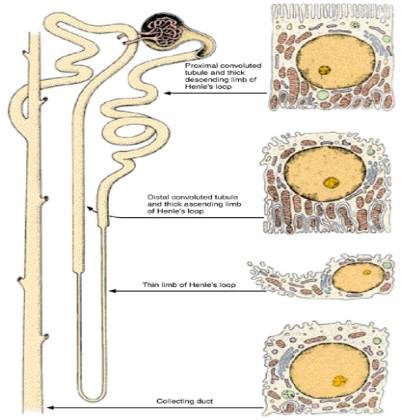 loop.
loop.
DISTAL CONVULUTED TUBULE: It is lined with cuboidal cells with a few µ-villi and some mitochondria.
COLLECTING TUBULE: It is lined with columnar epithelials. It contains 2 main types of cells, namely: -
a) Principal cells (P-cells or Light cells): They have a lighter cytoplasm, few granules, mitochondria and µ-villi. Long cilia projects in to the lumen.
b) Intercalated cells (I-cells or Dark cells): They have a darker cytoplasm, many granules, mitochondria and µ-villi. They are rich in carbonic anhydrase and H+-ATPase.
VASA RECTA: This is the portion of PTC around the loop of Henle in renal medulla that returns to cortex and empty in to veins. It plays a major role in counter current exchanger mechanism of urine formation.
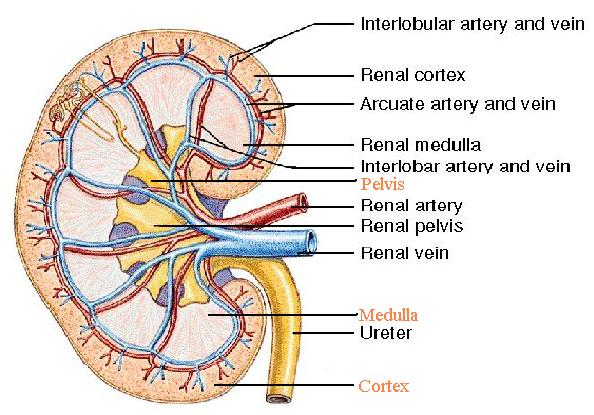
RENAL CIRCULATION: Abdominal aorta - Renal artery – Kidneys - Hilum (posterior – upper, middle, lower) - Hilum (anterior – apical upper, middle, lower) - Renal pyramids - Inter-lobar arteries - Arcuate arteries (base of pyramids) - Inter-lobular arteries - Afferent arterioles - Glomerular capillaries - Efferent arterioles – PTC.
[NOTE: From PTC, once again start replacing arteries with veins starting from inter-lobular and go up to renal vein]
MECHANISM OF URINE FORMATION
This process can be divided in to 3 steps, namely: a) Glomerular ultrafiltration, b) tubular selective reabsorption and c) tubular secretion.
1. GLOMERULAR ULTRAFILTRATION: It is a passive process in which water and solutes are filtered from the blood plasma and the filtrate goes in to Bowmann’s space.
Thus, Glomerular filtrate = Blood plasma – plasma proteins
The ultrafiltration occurs due to glomerular capillary pressure (75 mm Hg) which is opposed by oncotic pressure of plasma proteins (25 mm Hg) and intracapsular pressure (20 mm Hg).
Peff = Pg – (Pop + Pic). Thus, Peff = 75 – (25 + 20) = 30 mm Hg
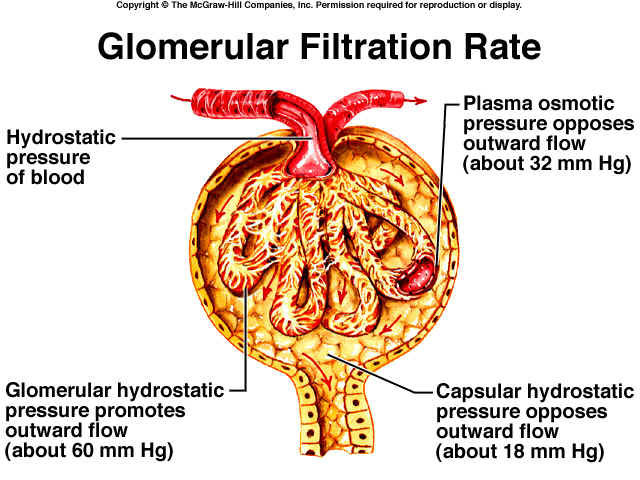
Generally, oncotic pressure of Bowmann’s capsule is not included in the above formula, because it is negligible.
Glomerular Filtration Rate (GFR) = Kf . Peff
Where, Kf is Filtration constant, which depends upon membrane permeability and total filtering area.
GFR = 125 mL/min (or) 180 L/24h; snGFR = 60 nL/min
GFR can be measured by inulin and creatinine clearance tests.
Filtration fraction (FF) = GFR/RPF = 125/650 = 0.19
Filtration fraction is the plasma fraction that is filtered. RPF is renal plasma flow rate.
FILTRATION BARRIERS
a) Mechanical/Structural: The glomerular basal membrane and to some extent the capsular epithelials provide barrier. This is due to the tiny pore size. Molecules up to 4 nm size can only be filtered.
b) Electrical/Charge: It is due to the – ve charge posed by the sulphated GAGs and other mucopolysaccharides present in the capsular well surface and basal membrane. Since, at blood pH 7.4, the plasma proteins are anionic and repulsion occurs.
FACTORS INFLUENCING GFR
|
GFR |
GFR |
|
^ glomerular capillary pressure |
^ Bowmann’s hydrostatic pressure |
|
^ area of filtering surface |
^ plasma oncotic pressure |
|
^ RPF rate |
- |
|
^ membrane permeability |
- |
2. SELECTIVE REABSORPTION
The volume of glomerular filtrate is 180 L/24h. But, the volume of urine output is only 1.5 L/24h. This is brought about by tubular selective reabsorption. PCT and loop of Henle are primarily concerned with reabsorption. This process may be either active or passive. The substances that undergo this process are classified in to 3 based on the extent of reabsorption, namely: -
TUBULAR MAXIMUM (Tm): Substances are actively absorbed entirely up to certain concentration. Above such concentrations, they are excreted. Eg: glucose, PO4 and amino acids.
GRADIENT TIME: If allowed to stay for a longer time, some substances are more reabsorbed. Eg: Na+.
REGULATION OF REABSORPTION
a) Obligatory reabsorption: Reabsorption is not concerned with regulating the levels of substances. Eg; water and NaCl (80%).
b) Facultative reabsorption: Reabsorption concerned with regulating the levels of substances. Eg: water and NaCl (20%).
TRANSPORT MECHANISMS – TYPES
A) PASSIVE (or) DOWNHILL: Movement of substances along the concentration or electrochemical gradient. Thus, no energy is required. It is of 4 types: a) simple diffusion, b) facilitated diffusion, c) bulk flow and solvent drag and d) non-ionic diffusion.
B) ACTIVE (or) UPHILL: Movement of substances against the concentration or electrochemical gradient. Thus, energy is required. It is of 3 types: a) 1o active transport, b) 2o active transport and c) counter transport.
GLOMERULO-TUBULAR BALANCE (GT balance)
It states that, ^ GFR = ^ Na+ reabsorption and decrease GFR = decrease Na+ reabsorption. This is a process which maintains Na+ balance and prevents excess loss or excess retention of Na+, when GFR ^ ses or ses. It occurs mainly in PCT due to 3 factors: a) PTC, b) blood factors and c) luminal factors. GT balance also occurs for other substances like glucose, HCO3-, etc.
[NOTE: The following table is a summarization of selective reabsorption. It will be easy to follow than reading individually for each substance]
|
SUBSTANCE |
AMT. FILTERED |
% TOTAL REABSORBED |
% REABSORBED IN VARIOUS REGIONS |
AMT. EXCRETED |
|||
|
PCT |
HL |
DCT |
CT |
||||
|
Glu |
125* |
~100 |
~100 |
- |
- |
- |
<100 mg |
|
AA |
- |
~100 |
~100 |
- |
- |
- |
<100 mg |
|
Ptns. |
- |
~100 |
~100 |
- |
- |
- |
<50 mg |
|
H2O |
125^ |
99 |
70 |
15 – 20 |
- |
10 – 15 |
1.5 L/d |
|
Na+ |
25000§ |
99 |
70 |
20 – 25 |
5 – 10 |
3 – 5 |
150§ |
|
K+ |
700 |
99 |
85 – 90 |
- |
- |
- |
90 |
|
Cl- |
18000 |
99 |
70 |
20 – 25 |
5 – 10 |
3 – 5 |
150 |
|
Ca2+ |
500 |
98 – 99 |
60 |
20 – 25 |
10 |
5 |
5 – 10 |
|
Mg2+ |
256 |
95 – 97 |
20 – 25 |
60 |
2 – 5 |
1 – 3 |
8 – 10 |
|
PO4- |
226 |
85 – 92 |
70 |
- |
- |
- |
23 – 32 |
|
HCO3- |
4300 |
~100 |
85 – 90 |
- |
4 – 5 |
2 – 3 |
- |
|
SO42- |
130 |
60 |
- |
- |
- |
- |
50 |
|
Uricate |
47 – 54 |
98 |
- |
- |
- |
- |
4 – 5 |
|
Urea |
900 |
(Varies) |
- |
- |
(Varies) |
- |
25 – 30 g/day |
[Key: * mg/day for organic substances; ^ mL/min for water; § mEq/day for ions]
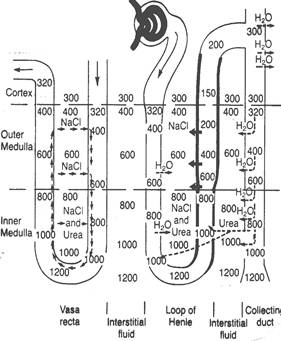
URINE OSMOLALITY & COUNTER-CURRENT MECHANISMS
Plasma = 280 – 295 mOsm/L. But, Urine = 500 – 800 mOsm/L. The ability of mammalian kidneys to excrete either concentrated or dilute urine is due to the counter-current mechanisms. It is a system in which the out-flow is close to, parallel to or counter to the inflow. It has 2 processes:-
a) COUNTER-CURRENT MULTIPLIER (CCM): It is an active system formed by Henle’s loop, especially the long loops of the juxtamedullary nephrons. There is an ^ ing osmolality of the interstitium from cortex to medulla.
F The glomerular filtrate in PCT and surrounding interstitial cells are isotonic w.r.t., plasma.
F
 A
progressive ^ se in osmotic gradient from the base to the tip of the
medulla is achieved.
A
progressive ^ se in osmotic gradient from the base to the tip of the
medulla is achieved.
F Thick ascending limb is impermeable to H2O.
F NaCl is actively transported out of the cell by Na+/K+/2 Cl- - pumps. This transport is not accompanied by H2O and thus, the tubule is hypotonic and interstitium is hypertonic.
F Thin ascending limb is also impermeable to H2O. NaCl moves out of the tubule passively.
F Thin descending limb is ^ ly permeable to H2O, but has low permeability to solutes (ions).
F H2O moves out of this limb in to the interstitium (hypertonic), making the tubular fluid hypertonic.
F Some solutes like urea also enter the descending limb which makes it more hypertonic.
F Thus, the tip of long Henle’s loop has the highest osmolality (1200 mOsm/L).
F DCT is relatively impermeable to H2O. Here, NaCl reabsorption is mediated by aldosterone, which makes the tubular fluid hypotonic.
F In collecting ducts also NaCl reabsorption is mediated by aldosterone, which makes the tubular fluid hypotonic.
F If ADH (vasopressin) is present, the entire length of CD becomes H2O-permeable thereby making the urine hypertonic. In the absence of ADH or improper functioning of ADH receptor, urine will be hypotonic (diluted and more volume).
ROLE OF UREA:
1. It contributes to medullary interstitial hypertonicity.
2. The inner medullary CD becomes ^ ly urea-permeable in the presence of ADH.
3. Urea then moves out in to interstitial space and causes hyperosmolality.
4.
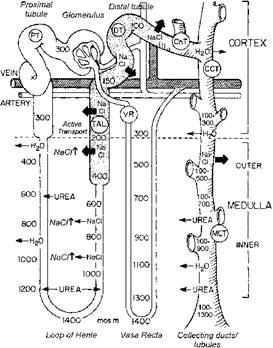 It
also enters the thin limbs and passes along DCT and CD to enter medullary CD in
^ concentration.
It
also enters the thin limbs and passes along DCT and CD to enter medullary CD in
^ concentration.
5. It also enters vasa recta and recirculates.
6. This medullary recycling contributes to about 40% of medullary osmolality when highly concentrated urine is formed. (diagram next page)
b) COUNTER-CURRENT EXCHANGER (CCE): It is a passive system formed by the vasa recta that ensures maintenance of osmotic gradient and prevents its dissipation.
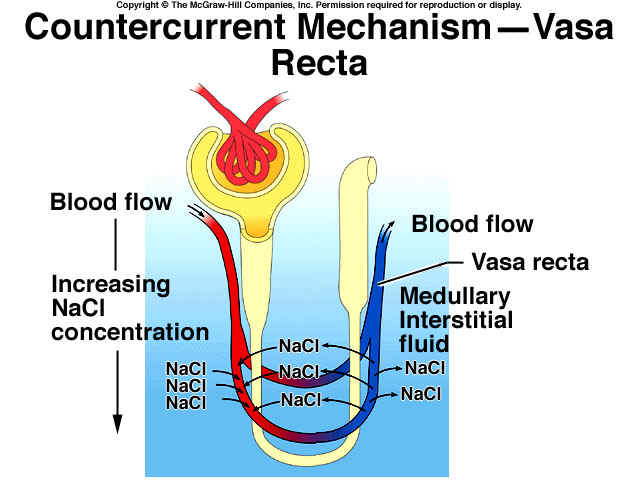
F Vasa recta (VR), which forms PTC loops close to HL, act as CC exchanger that maintains ^ [solute] as the blood flows in opposite direction in its 2 loops.
F The retention of excess solutes in medullary interstitial fluid is made possible by this CCE mechanism.
F The out-flowing blood tends to lose its solutes in to the inflowing blood.
F This recirculation in medulla retains both Na+ salts and urea that passively diffuses out of the ascending limb of VR in to the interstitium and from there to descending limb of VR.
F H2O, on the other hand, moves out of the descending limb in to the ascending limb to maintain hypertonicity.
TUBULAR SECRETION
Transport of substances from PTC in to the tubular lumen takes place. Na+, K+, H+, NH3, hippuric acid, ethereal SO4s, glucuronides, steroids, 5-OH-IAA are some examples.
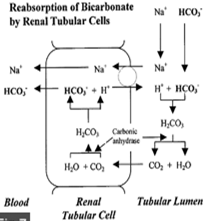
RENAL ACID – BASE REGULATION
Kidneys eliminate non-volatile fixed acids of about 40 – 80 mEq. They also control plasma HCO3- by reclaiming the filtered fraction by exchanging equal amount of H+. This adds to the acidity of urine.
Normal urine pH = 5 – 7 (ave); Extreme range = 4.5 – 8
 PLASMA
BICARBONATE BUFFER: HCO3-/H2CO3
is the major plasma buffer. The HH eqn. for this system is –
PLASMA
BICARBONATE BUFFER: HCO3-/H2CO3
is the major plasma buffer. The HH eqn. for this system is –
pH = pKa + log [HCO3-]/[ H2CO3]
pH = 6.1 + log [HCO3-]/0.03 x paCO2
where, paCO2 is 40 mmHg. Normal plasma [HCO3-] = 24 mEq/L
Thus, the ratio becomes 20 = log 20 = 1.3
Thus, pH = 6.1 + 1.3 = 7.4 (physiologic pH restored).
ACIDIFICATION OF URINE
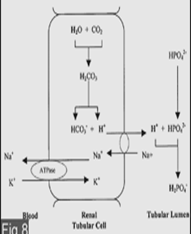 This
occurs mostly in CD. Most of the H+ secreted (98%) is used to reclaim
the filtered HCO3-. The H+ that is not utilized
for this process is excreted along with PO4 and NH3, which
replaces plasma HCO3-.
This
occurs mostly in CD. Most of the H+ secreted (98%) is used to reclaim
the filtered HCO3-. The H+ that is not utilized
for this process is excreted along with PO4 and NH3, which
replaces plasma HCO3-.
RENAL PHOSPHATE BUFFER:
[HPO42-]/[H2PO4-] is the chief renal buffer. Its HH eqn. gives the value,
pH = 6.8 + log [HPO42-]/[H2PO4-]
= 6.8 + log 4
= 6.8 + 0.6 = 7.4 (physiologic pH restored).
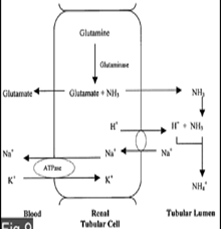
AMMONIA MECHANISM: At urine pH 4.5, the H+ gradient of tubular fluid ratio to that of plasma = 1000: 1. This prevents further H+ secretion. NH3, being lipid-soluble passes cell membrane by non-ionic diffusion in to the lumen and combines with H+ to form NH4+. Ammonium is non-diffusable. NH4+ combines with Cl- (90%) obtained from NaCl and excreted as NH4Cl. A small fraction is also excreted as (NH4)2SO4. The pK value of NH3/NH4 buffer is about 9.3
JUXTA GLOMERULAR APPARATUS & THE RENIN – ANGIOTENSIN SYSTEM
JGA is a specialized system composed of vascular and tubular components. The thick ascending Henle’s loop, on approaching its own glomerulus, comes in contact with the afferent and efferent arterioles.
JGA is present at this junction. It consists of 3 regions:- a) tunica media (secretes renin), b) macula densa; c) lacis cells. All these parts are innervated by sympathetic nerves.
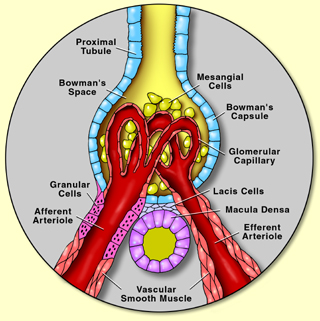 FUNCTIONS
OF JGA:
FUNCTIONS
OF JGA:
F It synthesizes and releases a glycoprotein hormone, renin that acts as a protease.
F It’s a α2 – globulin which converts angiotensinogen to AT – 1 by inducing angiotensin – converting enzyme. This enzyme is found in lungs, renals, encephalon and plasma.
F The JGA itself contains sensors which setup signal for renin release, viz., baroreceptors and chemoreceptors (NaCl levels).
ROLE OF R-A SYSTEM: (see diagram next page)
URINE – PHYSICAL CHARACTERISTICS
Volume: 1000 – 1500 mL/24h in healthy adult. This volume may vary due to diurnal variation, fluid intake, diet, temperature, body posture, exercise and emotion.
Colour: Normally amber or straw-yellow coloured due to urochrome.
Odor: Aromatic initially. But, becomes pungent on standing, due to NH3. Acetone gives sweet odor in ketosis patients.
Sp.Gr.: 1.015 – 1.022, which is proportional to solute concentration.
Osmolality: 500 – 800 mOsm/L. Extreme = 30 – 1400 mOsm/L. A rough estimate of osm can be obtained by measuring sp.gr.
pH: 5 – 7 (acidic in reaction). Extreme = 4.5 – 8. Alkaline on vegetarian diet and acidic on meat diet.
Sediments: Fresh urine is clear. Keeping it undisturbed for some time gives rise to flocculum that has nucleoproteins, mucoproteins, along with epithelial cells. Alkaline urine has Ca3(PO4)2, NH4 and MgPO4, oxalates and urates in addition.
CHEMICAL COMPOSITION OF URINE
|
H2O |
95 – 96% |
|
Inorganic |
mEq |
|
Na+ |
50 – 130 |
|
K+ |
20 – 70 |
|
Ca2+ |
5 – 10 |
|
P |
20 – 40 mmole |
|
Mg2+ |
5 – 15 |
|
Cl- |
50 – 130 |
|
Titratable H+ |
15 – 30 |
|
SO42- |
30 – 45 |
|
NH3 |
20 – 40 |
|
ORGANIC |
g |
|
Urea |
10 – 20 |
|
Urea N2 |
5 – 10 |
|
Creatinine |
0.7 – 1.5 |
|
Uric acid |
0.2 – 1.0 |
|
Acids |
10 – 25 mmole |
URINE – ABNORMALITIES
This may be either due to renal disorders or non-renal causes. They are:-
a) Volume: Polyuria (2.5 L/d); Nocturia (^ at night); Oliguria (≤ 400 mL/d); Anuria (≤ 100 mL/d).
b) Appearance: High-coloured (dehydration, fever); light-coloured (diuresis); cloudy (precipitation of solutes and mucous); turbid or smoky (hematuria, pus or chyluria); high-yellow (jaundice); brown (hemoglobinuria).
c) Abnormal constituents
1. Physiological proteinuria: due to severe exercise, high protein meal, pregnancy.
2. Pathological proteinuria: due to renal diseases, heavy-metal poisoning, inflammation, chronic renal failure. Eg: Bence-Jones proteins occurring during Hodgkin’s disease, leukemia, lymphosarcoma, etc.
3. Glycosuria: usually associated with glucose (> 1 g/d). Others such as fructose, galactose, lactose and pentosuria can also occur.
4. Ketone bodies: due to ^ metabolic ketosis (> 3 – 15 mg/d). Acetone which usually occurs can be detected by Rothera’s test.
5. Hematuria: RBC in urine due to glomerulonephritis or urinary tract bleeding.
6. Hemoglobinuria: Hb in urine due to intravascular hemolysis.
7. Bilirubin: during jaundice.
8. Creatinuria: this compound is usually absent in healthy adult men. It usually occurs in small amount in children, normal women and also post-partum. A high level is an indicative of muscle degenerative disorders.
9. Porphyrins: Normally coproporphyrin (60 – 280 mg/d) is present. Uroporphyrin and ^ coproporphyrin occur in acute porphyria. Coproporphyrin-III is excreted in lead poisoning.
10. Amino aciduria: homogentisic acid (alkaptonuria), C, K, R, ornithine (cysteinuria and Fanconi’s syndrome).
d) Casts: collection of pptd. proteins. It consists of proteins only. Other types of casts include hyaline casts, blood casts and granular casts. It is used to diagnose glomerulonephritis.
v DIURETICS: These are chemical agents that ^ se urine volume and output which is known as diuresis. A no. of drugs that acts as diuretics in different ways at different sites and is used to treat edema and hypertension. (table next page).
|
CLASS |
MODE OF ACTION |
COMPOUNDS |
|
Loop diuretics |
Θ NaCl reabsorption in thick ascending limb |
Furosemide, ethacrynic acid |
|
Thiazide diuretics |
Θ NaCl reabsorption in thick ascending limb and DCT. They have longer duration of action and produce less severe diuresis |
Chlorothiazide |
|
Mercurial diuretics |
Θ NaCl reabsorption in thick ascending limb. Administered i.v. only. Toxic and not used now |
Mersalyl |
|
Aldosterone antagonist |
Θ NaCl reabsorption and K+ excretion in cortical CD |
Spironolactone |
|
Amiloride |
Θ NaCl reabsorption and K+ excretion in cortical CD. But this is not a direct antagonist of aldosterone |
Amiloride |
|
Carbonic anhydraseΘ rs |
Θ HCO3- reabsorption and H+ secretion to produce alkaline urine |
Acetazolamide |
|
Salts |
Not strictly a diuretic. Usually used to change urine pH |
Acidifying salt (NH4Cl); alkalinizing salt (K-citrate) |
|
Alkaloids |
^ GFR and decrease NaCl reabsorption |
Caffeine, theophylline |
|
Alcohol |
Θ ADH secretion |
Ethanol |
RENAL DISORDERS
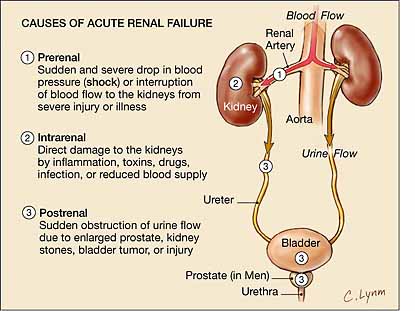
Reduction in renal mass by ½ does not produce any disturbance in normal renal function. A further reduction to < 40% causes functional impairment. A mild reduction in function is known as renal insufficiency. A severe reduction is known as renal failure. 2 types of renal failure are acute (ARF) and chronic (CRF).
|
ARF |
CRF |
|
Rapid, severe, reversible |
Gradual, severe, irreversible |
|
CAUSES |
|
|
Acute glomerulonephritis, acute tubular necrosis (ischemia) |
Chronic glomerulonephritis, diabetic nephropathy, urinary tract obstruction |
v ARF: It can be divided in to 4 stages:-
1. Circulatory insufficiency, peripheral and renal vasoconstriction, release of epinephrine, ADH, cortisol, ACTH and GH.
2. Afferent arteriolar vasoconstriction due to sympathetic over-activity. This leads to low GFR and thus, low Na+ filtration.
 [NOTE:
At this stage, urinary output can be rectified, when circulatory insufficiency
is corrected]
[NOTE:
At this stage, urinary output can be rectified, when circulatory insufficiency
is corrected]
3. This stage is known as incidient acute oliguric renal failure due to ^ sympathetic over-activity with severe vasoconstriction.
4. Established ARF due to cellular degeneration in basal lamina and acute tubular necrosis.
v CRF: Also known as uremia. It occurs when renal function falls to ≤ 25% (when ≥ 70% of nephrons have lost their normal functioning). Notable features are:-
a. Low GFR = 5 mL/min (chronic dialysis is needed).
b. Oliguria occurs which later develops to anuria.
c. Retention of N2 wastes in blood known as azotemia. This condition leads to uremia.
d. Retention of Na+ and H2O (edema).
e. Hyperkalemia.
f. Acidosis due to decreased H+ secretion, decreased NH3 formation and decreased HCO3- absorption.
g. Decreased erythropoietin = anemia = breathlessness.
h. Decreased calcitriol – low Ca2+ absorption – bone malfunctions.
i. ^ renin = ^ hypertension.
j. Gastrointestinal disturbances.
k. Neurological disturbances due to N2 metabolites.
l. Bleeding disorders due to thrombocytopenia and platelet defects.
m. ^ peptide hormone levels in blood.
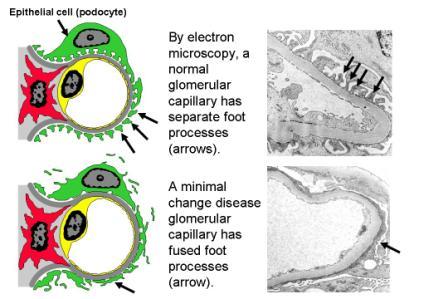
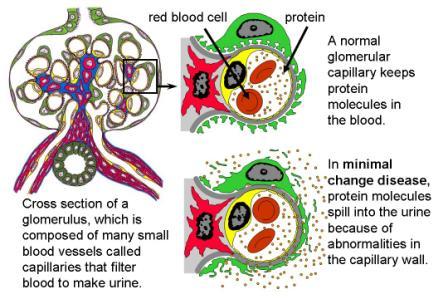
NEPHROTIC SYNDROME: arises due to ^ glomerular permeability that leads to ^ proteinuria, hypoproteinemia and edema. It is associated with minimal change disease.
DISORDERS OF RENAL TUBULAR TRANSPORT
Usually these are congenital, resulting from a decrease in activity or abnormality of concerned genes. They are:-
1. Renal glycosuria
2. Hartnup’s disease, Fanconi’s syndrome (defect in amino acid transport proteins leading to amino aciduria, glycosuria, ^ phosphaturia, renal tubular acidosis, ^ hyperkaluria)
3. Renal tubular acidosis (low H+ secretion - ^ HCO3- loss)
4. Decreased PO4 reabsorption leads to vit-D – resistant rickets
5. Pseudohypoparathyroidism (inability to ^ se phosphaturia in response to PTH – PO4-emia – low Ca2+-emia)
6. Nephrogenic diabetes insipidus (inability of CT to respond to ADH)
7. Pseudohypoaldosteronism (inability of DCT to respond to aldosterone)
8. Bartter’s syndrome (^ hyperkaluria, polyuria, low kalemia, ^ plasma aldosterone, hyperplasia of JGA, but, normal BP due to insensitivity to AT-II)