MUSCLES are the type of tissues that have developed the maximum ability to contract, which is the fundamental property of protoplasm. 3 types of muscles are:
1) SKELETAL/VOLUNTARY/CROSS-STRIATED/STRIATED: Innervated by somatic nerves that is essential for their contraction. (Except intercostal muscles in respiratory system that are involuntary).
2) CARDIAC/INVOLUNTARY/PARTLY STRIATED: Contracts even in the absence of innervations. They are under control of ANS that regulates them.
3) SMOOTH/PLAIN/INVOLUNTARY/UNSTRIATED: Present in viscera, blood vessels, etc. They are under control of ANS that regulates them. Contracts even in the absence of innervations. (Except urinary bladder, anal sphincter, etc that are partly voluntary).
3 TYPES OF MUSCULAR MOVEMENTS
a) Locomotion, b) Movement of body parts and c) Movement of substances with body. [a & b involves skeletal while c involves smooth].

SKELETAL MUSCLE
They form the flesh. 40% of total body wt. Contain about 1/3rd of body proteins (protein reserve) and contributes to about half of the body metabolism. They also serve as glycogen reserve. Water (75%), Solids: proteins (20%), others (5%).
Muscle fibre, which is a long cylindrical multinucleated cell, is the structural and functional unit of a muscle. A large number of muscle fibres constitute the skeletal muscle.
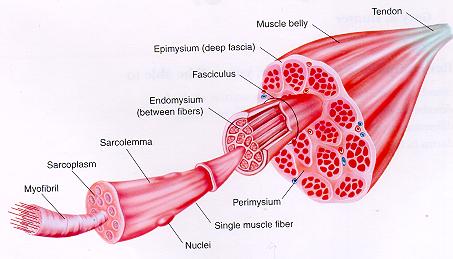
The fibres do not branch, nor anastamose with adjacent fibres.
Length of fibres varies from 1-100 mm (ave: 30-50 mm) depending whether it is long or short.
Some fibres run from end to end of a muscle (e.g.: sartorius), but mostly they are short.
Diameters vary from 10-100 μm (ave: 50 μm).
The ends of the individual fibres are attached to the interlacing connective tissue network within the muscles.
The ends of a muscle are attached to the bone by collagen fibres, known as tendons.
The contractile forces of the individual fibres are transmitted through the collagen of parenchymatous connective tissue and collagen fibres of tendons to the bones.
FUNTIONS OF CONNECTIVE TISSUE INVESTMENT
Ø Blood vessels run along these septa around each muscle fibre.
Ø Binds the contractile units and integrate their activity.
Ø Maintains individuality of each muscle fibre and facilitates their movement.
FIBRE TYPES IN SKELETAL MUSCLE
|
RED/SLOW FIBRE |
WHITE/FAST FIBRE |
|
Smaller, narrower, more opaque, thick Z disks, more mitochondria, sarcoplasm, myoglobin and oxidative enzymes |
Translucent, less sarcoplasm, mitochondria, cytochrome, rich in phosphorylase, thin Z-lines |
|
Cross striations less marked |
Prominently marked |
|
Motor unit contains more fibres |
Fewer fibres |
|
Contracts slowly, longer latent period, tetanized at slower rates of stimulation |
Rapid contraction, tetanized at higher frequency of stimulation |
|
Fatigued less easily |
More easily |
|
Higher O2 store, capillary network, activity of myosin-ATPase lowest |
Lower O2 store, capillary network, activity of myosin-ATPase highest |
|
Aerobic type metabolism. Less energy expense due to low ATPase activity |
Aerobic type metabolism. More energy expense due to high ATPase activity |
|
Can tolerate oxygen debt and sustain prolonged contraction |
They show short bursts of activity which is never sustained |
CONTRACTILE PROTEINS
1.
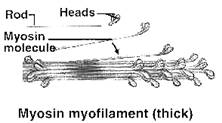
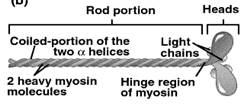 MYOSIN:
(M.wt. 400-800 kDa)
MYOSIN:
(M.wt. 400-800 kDa)
· 35% of total muscle proteins and one of the main contractile elements.
· Cleaved by proteases like trypsin to two components: light meromyosin (100 kDa) from rod-like portion and heavy meromyosin (200 kDa) from globular portion.
· Myosin has tendency to form dimer and show slow birefringence (different refractive index due to electrical oscillation).
· Cleavage by papain gives off two heavy meromyosin from the globular end, a part of which is in α-helical form.
· ATPase activity is linked with heavy meromyosin.
 (Heavy
meromyosin + ATP Phosphomyosin + Actin Actomyosin complex)
(Heavy
meromyosin + ATP Phosphomyosin + Actin Actomyosin complex)
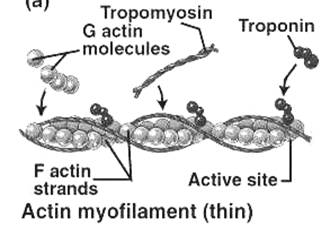
2. ACTIN: (M.wt. 43 kDa)
· 14% of total muscle proteins. Extracted by 0.6 M KI solution.
· G-Actin: 57-70 kDa. Monomeric globular form. Forms complex with ATP in solution.
· F-Actin: Double-helical, fibrous, polymeric form. Forms complex with ADP.
F-actin is present in muscles and has binding site for myosin-head. It has no ATPase activity and no cross-bridges. G-actin polymerizes to F-actin in the presence of salts.
3.
 TROPOMYOSIN
(M.wt. 70 kDa)
TROPOMYOSIN
(M.wt. 70 kDa)
6-10% of total muscle proteins. Join end to end to form long filaments. Crystallizes in 90% water and form lattice. Has two helical strands wound around each other. Located in groove between actin strands (each molecule extends over 7 actin). During resting state it appears to cover the binding site of actin to myosin.
4. TROPONIN [TN] (M.wt. 50 kDa)
Globular unit located at intervals along tropomyosin strands. 3 sub units:
(i) TN-C – have affinity for Ca2+ and have binding site for it.
(ii) TN-T – binds to tropomyosin and binds the other two sub units of it.
(iii) TN-I – small molecule with high affinity for actin and binds to it.
(M.wt. of these subunits vary from 18-35 kDa).
Each actin filament = 300-400 actin + 40-60 tropomyosin + 40-60 troponin (ratio 7:1:1). F-actin is attached to Z-line by a protein α-actinin. Tropomyosin and troponin are regulatory contractile proteins because they interfere with actin-myosin interaction.
FINE STRUCTURE OF A MYOFIBRIL
1. Each muscle fibre contains numerous oval nuclei situated superficially under the Sarcolemma.
2. The cytoplasm is called Sarcoplasm, which surrounds a number of closely packed fine thread-like structures called Myofibrils (about 1000 in each fibre).
3. Each myofibril is about 1-2 μm diam. and runs end to end of fibre appearing as fine parallel lines.
4. These myofibrils in turn contain Myofilaments (miscellae) that contain the contractile proteins. They are of 2 types:-
v THICK OR MYOSIN FILAMENT: Contains myosin and filament diameter is 10-12 nm and length 1.5 or 1.6 μm.
v THIN OR ACTIN FILAMENT: Contains actin, tropomyosin and troponin. Diam. 5-6 nm, length 1 μm.
v Each thick filament is encircled by 6 thin ones (hexagonal arrangement), i.e., each actin filament is shared by 3 myosin filament.
5. When viewed under a light microscope, we can see a) longitudinal striations due to parallel myofibrils and b) transverse or cross-striations due to alignment of alternating bands of different refractive indices (μ), giving rise to dark and light regions.
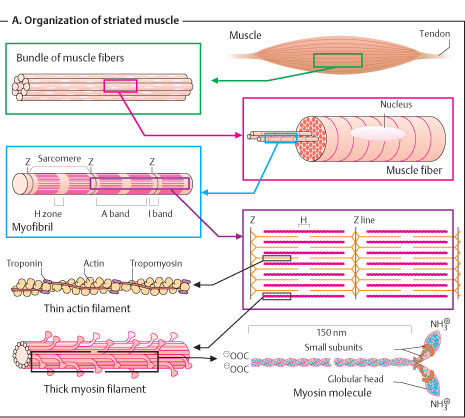
6. A-BAND: Anisotropic band. Appears dark because of high μ and is birefringent. Made of thick filaments and parts of thin filaments that overlap the thick ones. Length-1.5 μm.
7. H-BAND: Hensen’s band. It is in the middle of A-band where the actin filament does not overlap with myosin filament. This region is isotropic and appears light.
8. M-LINE: Myosin line that is formed by cross connections of myosin filaments in the middle of H-band.
9. I-BAND: Isotropic band. Appears light because of low μ. Made of thin filaments that do not overlap the thick ones. Its length in average is 1 μm.
10. Z-LINE: Dobie line/Z-disk. Made of fine filaments that connect actin filaments in the middle of the I-band and appears dark relatively.
11. SARCOMERE: It is the part between two adjacent Z-lines and is the ultimate contractile unit. Length is usually 1.5-3.5 μm but varies upon the state of contraction and stretch.
THE SARCOTUBULAR SYSTEM
Muscles contain 2 major systems of tubules which play an important in the excitation of muscle and in coupling of excitation with contraction.
1. TRANSVERSE TUBULAR SYSTEM (T-TUBULES/T-SYSTEMS):-
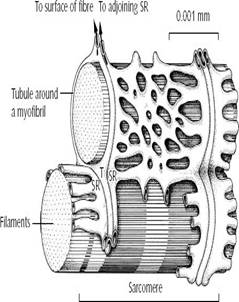
Ø These tubules (diam-0.03μm) are continuous with the sarcolemma and extend inwards from the surface of muscle fibre and form a ring around every fibril, termed annulus.
Ø This is an extension of EC space in to the fibre and contains the interstitial fluid.
Ø In mammalian skeletal muscle it is present at the junction of A and I-bands. (In cardiac muscle it is present near Z-disk).
Ø They function to rapidly transmit the action potential from the surface membrane to the deeply placed muscle fibres.
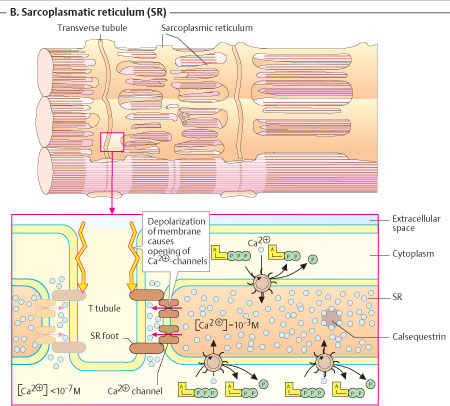
2. SARCOPLASMIC RETICULUM (SR):-
Ø Longitudinal system of tubules (diam-0.04μm) that run parallel to muscle fibres.
Ø Their ends broaden to form large sacs called terminal cisternae (stores Ca2+) which lie close to annulus of the T-tubules.
Ø This cluster of T-annulus and two terminal cisternae of SR constitute the Muscle Triad.
Ø The terminal cisternae form long narrow tubes which broaden at H-zone.
Ø Ca2+ is released when action potential reaches annulus.
Ø It is then pumped back in to the central portions of the tubules from where it moves to cisternae
NEUROMUSCULAR JUNCTION AND TRANSMISSION
Skeletal muscles normally contracts only as a result of impulses transmitted from motor neurone to the muscle across the myoneural junction.
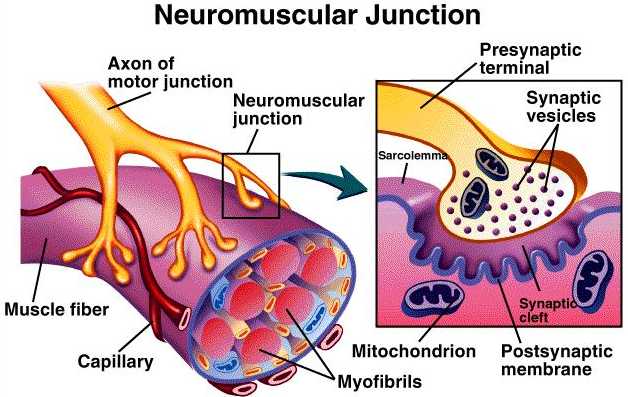
Each end foot contains about 300,000 vesicles and each vesicle contains up to 10,000 Ach molecules.
Region of motor end plate under sarcolemma contains mitochondria, nuclei and Ach esterase that split up Ach to acetate and choline.
The end foot also contains the enzyme Ach synthetase that forms Ach from acetyl-CoA and choline.
NERVE SUPPLY TO MUSCLE
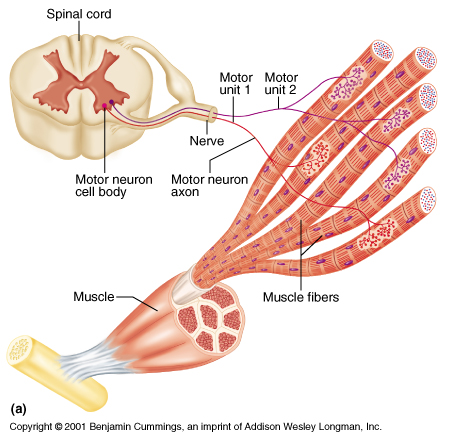
§ Muscle spindles: These are sensory end organs (receptor organs) found between contractile muscle fibres.
§ Skeletal muscles are innervated by somatic nerves, most of which are efferent.
§ The efferent nerve is the motor neurone.
§ The motor nerve fibres are large myelinated with cell bodies in ventral grey matter of spine.
§ Each of them gets divided in to about 150 (range: 7-1700) branches and each branch ends in a muscle fibre by a special end organ called motor end plate.
§ The synapse between terminal branch of motor nerve and muscle fibre is called myoneural junction or NM junction.
§ There are also afferent nerve fibres, mostly non-sensory, from muscles spindles and tendon receptors which convey information about state of stretch of muscle.
§ A few nerves convey pain sensation.
MECHANISM OF SKELETAL MUSCLE CONTRACTION

(Note: These steps are similar to synaptic nerve transmission where instead of a post synaptic neuron we have a muscle fibre)
Mg2+ inhibits Ach release from vesicles. Hydrolysis of Ach at motor end plate prevents continuous response to one stimulus.
Miniature End Plate Potential (MEPP): In the resting state of NM junction, small quantities of Ach are released randomly (1 Ach/sec) which causes a very small non propagated depolarization of about 0.5 mV. This gives rise to MEPP.
THEORIES ON MECHANISM OF MUSCLE CONTRACTION
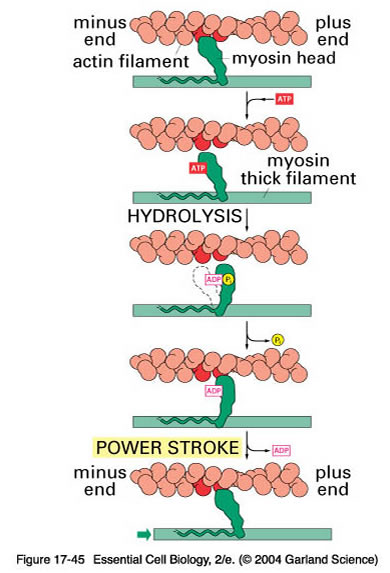
THE SLIDING FILAMENT THEORY (CROSS-BRIDGE THEORY)
According to this theory deduced by Hugh Huxley (1954), the actin filament (A) slides towards the myosin (M) filament. The myosin head link with the reactive sites of actin filament, bend and pull it inwards.
A. THE RESTING STATE:
1) Tropomyosin-TN complex prevents interaction of M and A filaments.
2) Reactive sites of A are negatively charged and the M cross-bridges (that are attached to ATP) is also negatively charged (in the absence of Ca2+) and so repel and kept apart.
3) Binding site of actin is also covered by tropomyosin.
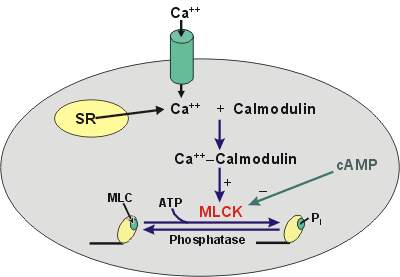
4) Only on availability of Ca2+ both filaments binds together (- ve charge neutralized by Ca2+).
5) M develops ATPase activity.
6) Ca2+ initiates contraction by uncovering the binding site of actin.
B. SEQ. OF EVENTS DURING CONTRACTION
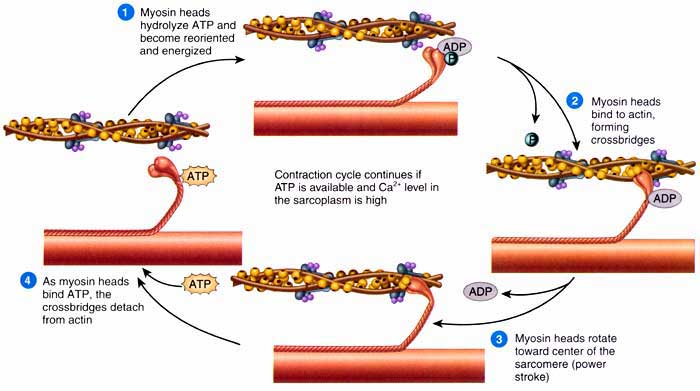
The whole process explained below is known as Excitation-Contraction Coupling.
1) AP from nerve fibre reaches NM junction and transmitted to muscle fibre
2) AP in muscle spreads along sarcolemma
3) AP spreads inwards through T-tubules and reaches annulus of triad
4) Ca2+ release from terminal cisternae of sarcoplasmic reticulum
5) Ca2+ diffuses towards myofilaments and initiates contraction
6) Ca2+ binds to TN-C causing TN-I binding with actin weakened
7) Uncovering of binding sites of actin
8) Head of M links to A, and then moves inwards by bending which pulls the A inwards
9) The 2 A of sarcomere move in opposite direction, i.e., towards the centre.
10) The bend in M head exposes ATP binding site and allows ATP to come and attach
11) This causes detachment of M head from A and ATP is split consequently (stimulated by Ca2+)
12) Ca2+ is then pumped again in to SR and stored in cisternae. This causes muscle relaxation
Energy for transfer of Ca2+ in to SR is also provided by ATP. Thus ATP is involved in both contraction and relaxation (plasticizer action) of muscle.
2 TYPES OF CONTRACTION
1. ISOTONIC: Involves actual shortening of muscle. It has 3 phases:
a) Latent period (0.01 s) – no apparent change.
b) Contraction phase (0.04 s)
c) Relaxation phase (0.05 s)
2. ISOMETRIC: There is not change in length of muscle on changing tension.
THERMAL CHANGES DURING MUSCLE CONTRACTION
Resting muscle liberates heat at a constant rate, known as basal heat. Increased amount of heat is given off during contraction, relaxation and after relaxation by the muscle. It may be divided in to:
a. INITIAL HEAT: Heat given off during twitch even under anaerobic conditions. It consists of:
I. Heat of activation: Occurs when muscle passes from resting to active state. This is delivered at a rapid rate even before the mechanical process of shortening takes place. A number of such heats get summed up on receiving a number of stimuli to form what is called maintenance heat.
II. Heat of shortening: Liberated when muscle shortens and thus proportional to it. Present only during isotonic contraction.
III. Heat of relaxation: As the muscle lengthens during relaxation, the work done during contraction is liberated as heat. (I and II are the waste heat of chemical reactions).
b. DELAYED HEAT: Also known as recovery heat that continues for several minutes after mechanical events are over. This is due to metabolic processes which restore the muscle to its resting state. This value is lesser in anaerobic (delayed anaerobic heat) state than in aerobic state (oxidative delayed heat).
CHEMICAL CHANGES DURING MUSCLE CONTRACTION
Muscle contains a number of high energy phosphate compounds along with biomolecules that contribute for its energy.

5. FFA: Used as fuel that gives out ATP only during resting and light work conditions.
O2 DEBT: During severe exercises of short duration, the muscle contracts in the absence of O2. So, under this anaerobic state, lactate is formed from pyruvate. The extra O2 required to oxidize lactate is made available after he exercise. This portion of O2 is the O2 debt.
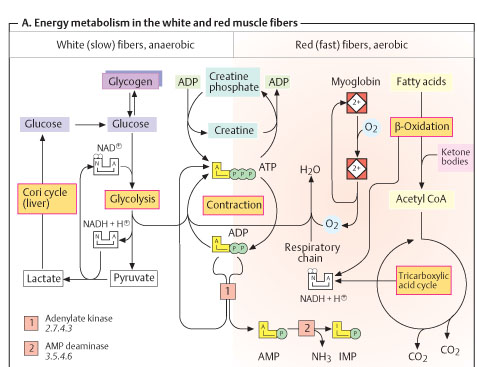
CORI CYCLE: When large amount of lactate is produced it diffuses in to blood and gives out the above reaction. 1. Glycogenolysis followed by anaerobic glycolysis, 2. Gluconeogenesis followed by glycogenesis, 3. Aerobic glycolysis, 4. Glycogenesis.
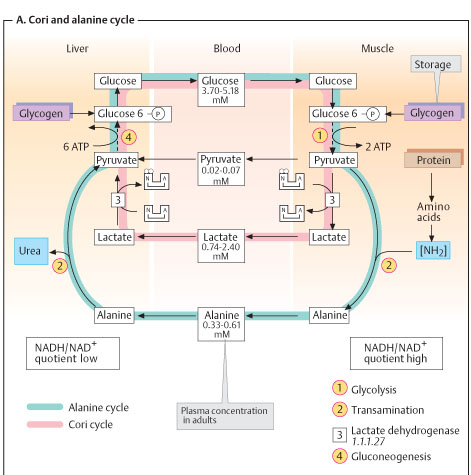
pH CHANGES DURING MUSCLE CONTRACTION
ATP hydrolysis causes fall in pH. Creatine formation raises pH. Later, lactate formation decreases pH when salts, protein and CP act as buffers. Prolonged contraction also liberates NH3 which serves to maintain reaction.
RIGOR MORTIS (post mortem rigidity)
All muscles after removal from the body or after death undergo changes in the protoplasm associated with permanent loss of irritability. This is rigor mortis. A living muscle fibre has its specific length, it is translucent, elastic, extensible, soft and alkaline (pH 7.3). In rigor mortis, it becomes shorter, opaque, inelastic, inextensible, rigid and acidic (pH 5.8). The proteins get coagulated.
SKELATAL MUSCLE FIBRES – SOME PROPERTIES
1) ALL-OR-NONE PHENOMENA: This property is exhibited by an individual muscle fibre. When a muscle fibre is stimulated, it will show maximal contraction irrespective of the intensity of stimulus. So, minimal contraction means a few muscle fibres are involved in a muscle, whereas a maximum contraction means all the muscle fibres are involved.
2) RHEOBASE: It is the strength of the weakest galvanic current which will excite a muscle or a nerve when allowed to flow for a sufficient duration of time (utilization time).
3) CHRONAXIE: The shortest duration of time that a current of twice the rheobasic strength must flow in order to excite a muscle or a nerve. Shorter the chronaxie value greater will be the excitability of muscle.
4) CONTRACTILITY: Ability to contract (develop tension).
5) EXCITABILITY (IRRITABILITY): Ability to respond to a stimulus either electrochemical or mechanical (blow to muscle by external force).
6) EXTENSIBILITY: Ability to be stretched.
7) VISCOELASTICITY: Having the ability to stretch or shorten over time.
8) ELASTICITY: Ability to recoil to normal length.

SMOOTH MUSCLES - STRUCTURE
§ Spindle-shaped. Each fibre has central oval nucleus. Average length 50-200 μm and diameter 2-10 μm.
§ Some muscle fibres may be as small as 15 μm in small arterioles and as large as 500 μm in pregnant uterus.
§ Only longitudinal striations and no cross striations.
§ This is because of no proper arrangement of actin and myosin (they both are present in a different isoform).
§ Tropomyosin is present, but TN-C is absent. Instead it has Ca2+ binding protein called Calmodulin.
§ There are no Z-line and T-tubules. SR is poorly developed.
§ Fewer mitochondria are present.
CHARACTERISTICS
§ Smooth muscles are generally not under voluntary control, except a few like urinary bladder and anus which have limited voluntary control.
§ They are innervated by both sympathetic and parasympathetic nerves where both perform antagonistic function.
§ They can also contract in the absence of innervation. Thus, nerves regulate contraction rather initiating it.
§ Denervation does not cause degeneration of muscle.
§ They are sensitive to chemical stimuli.
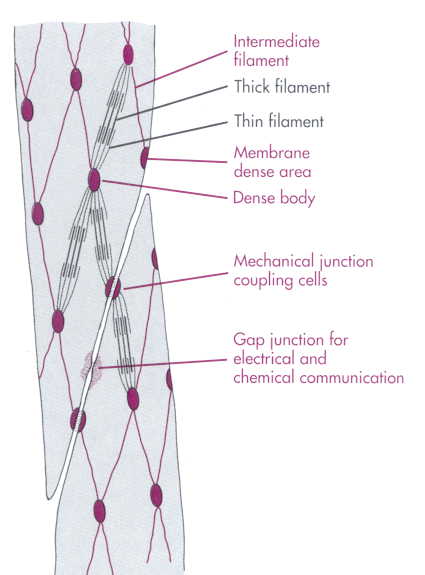
TYPES 1. VISCERAL/UNITARY:
§ Large sheets of closely packed muscle fibres.
§ Tight junctions between adjacent cells have low electrical resistance (high conductance).
§ There is no protoplasmic continuity between the cells.
§ They function as a syncitium.
§ Present in walls of hollow viscera (GI tract, uterus, ureter and UB).
§ Innervation is scarce and each individual fibre may not receive its nerve terminal.
§ Individual units of muscle fibres each receive a nerve terminal.
§ Impulse does not spread to adjacent fibres and is non-syncitial.
§ Present in iris and ciliary body of eye, pilomotor muscles.
§ Contractions are discrete, fine and localized which need innervation.
§ They also respond to chemicals and generally activated by Ach and NE.
§ NE mediated stimulus causes sustained response that resembles tetanus, due to repeated firing.
[Note: Blood vessels contain both multi-unit (large arteries) and unitary (small arterioles) smooth muscles]
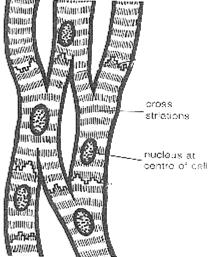
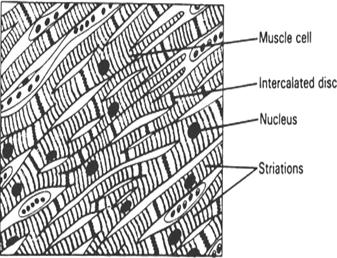
CARDIAC MUSCLE - STRUCTURE
SMOOTH MUSCLE CONTRACTION