This is a prominent, spherical or oval structure found at the centre of the cell. It is the controlling centre of all cell activities and has been described as the brain of the cell or controlling unit of the cell. It was first discovered by Robert Brown in 1831 in flowering plants. The study of nucleus is termed as karyology. It regulates all metabolic and hereditary activities of the cell.
Occurrence:
The nucleus is present in all eukaryotic cells. However, it is absent in erythrocytes and some lens cells. In eukaryotes, the nucleus is surrounded by a nuclear membrane whereas in prokaryotes, the nuclear material is not surrounded by a nuclear membrane. Such a nuclear material is called as nucleoid.
Number:
Generally a cell contains only one nucleus. But sometimes tow or more nuclei are present. Based on the number of nucleus, the cells are classified into the following types:
Anucleate cell – In anucleate cells the nucleus is absent eg. Erythrocytes of human
Mononucleate cell – In mononucleate cells a single nucleus is present ef. Amoeba
Binucleate cell – In binucleate cell, two nuclei are present. Of these nucleus is small called micronucleus and the other nucleus is large called macronucleus. Eg. Paramecium
Multinucleate cell – It contains many nuclei. Eg. Opalina
Position:
The position of the nucleus in a cell is variable. Usually it is situated in the centre of the cell. But in adipose cells or in egg rich in yolk, the nucleus is forced to lie on the periphery. In glandular cells and in Acetabularia it lies in the basal region.
Shape:
The shape of the nucleus varies considerably. In most of the cells it is spherical in shape. In cylindrical cells it is elliptical. In human neutrophils it is trilobed. In paramecium, the macronucleus is kidney shaped. The nucleus of spinning gland cells of insects is highly branched. In vorticella it is horse-shoe shaped.
Size:
Size of the nucleus is variable. The size of the nucleus is directly proportional to the cytoplasm. The more the volume of the cytoplasm the larger is the size of the nucleus. R.Hertwig has formulated a relationship between the nuclear volume and the cytoplasmic volume which is called the nucleoplasmic index (NP). The NP ratio acts as a stimulus to the cell division.

The size is also correlated with the number of chromosomes and the DNA content. The nuclei having triploid and tetraploid sets of chromosomes are larger is size than those having diploid sets of chromososmes.
NUCLEUS STRUCTURE:
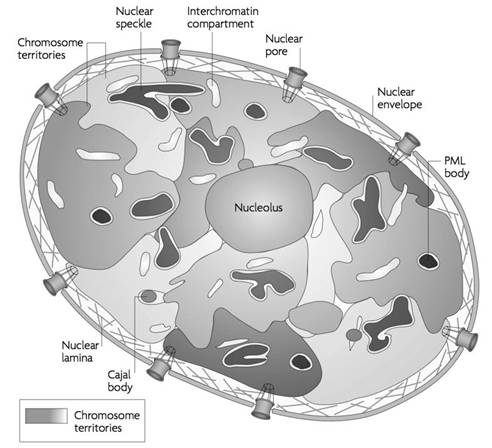
The nucleus is composed of the following structures:
This is a double-layered membrane which separates the nucleoplasm from the cytoplasm. The nuclear membrane has minute pores which allow the selective transfer of material between the nucleoplasm and the cytoplasm. The outer layer is called ecto karyotheca and the inner layer is called endo karyotheca. They are separated by a perinuclear space which is about 150 – 300Ao. Each outer and inner layer is about 70 – 80Ao thick. The outer membrane is often continuous with membrane of the golgi, endoplasmic reticulum, mitochondrion and plasma membrane. The outer membrane is rough due to the presence of ribosomes, while the inner membrane is smooth.
Within the nuclear membrane, completely filling up the space is a clear, semi-solid, granular substance or matrix called the nucleoplasm. The nucleolus and the chromatin network lie suspended in the nucleoplasm. The nucleus is filled with a homogenous, transparent acidophilic substance known as the nuclear sap or karyolymph. There are one or more definite structures called nucleoli. The chromatin threads remain suspended in the nucleoplasm. In addition there may be larger bodies which stain like chromatin thread and hence they are know as chromatic nucleoli or false nucleoli. The nulear sap contains organic and inorganice substances like nucleic acids, prtoteins, enzymes and minerals.
This dense, spherical granule found in the nucleus contains RNA (ribonucleic acid) which is responsible for protein synthesis in the cytoplasm.
These are very fine thread-like, coiled filaments uniformly distributed in the nucleoplasm. At the time of cell division, the chromatin becomes thick and ribbon like and are known as chromosomes. The chromosomes contain genes, which are composed of DNA (deoxy-ribonucleic acid). Genes are responsible for storing and transmitting hereditary characteristics from one generation to another. A gene is the functional unit of a chromosome. Genes are arranged in single linear order along the chromosome. One gene may be responsible for a single characteristic, or a single characteristic may be transmitted by a set of genes.
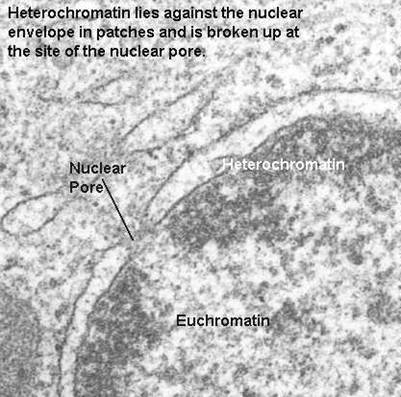
This network readily stains with basic dyes. The chromatin network is condensed to form thick ribbon-like bodies called chromosomes during cell division. At certain places, the chromatin net remains condensed as darkly stained chromatin mass. These regions are called as heterochromatin. They contain small amount of DNA and large amount RNA. At cerain stages of cell division, the chromatin reticulum may show bead-like structures called chromomeres. Nucleus appears differently during interphase and dividing phase (mitotic phase). During interphase, nuclear material appears as chromatin form whereas in mitotic phase it appears as chromosome form.
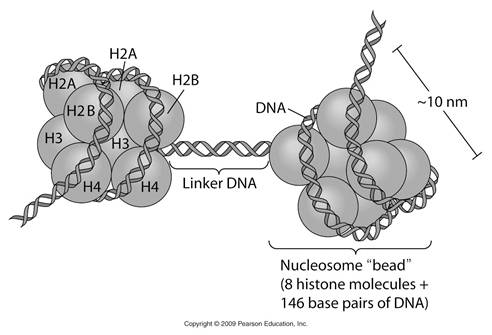
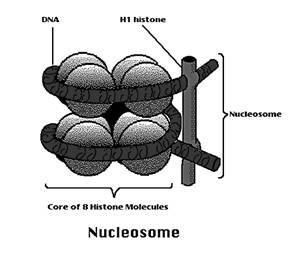
Chromatin is nothing but the knotted thread which contains nucleosome joined by linker DNA. In chromatin double helix DNA wounds around the histone core to form a super coiled nucleoprotein fibre. The DNA wraps around the histones to form a bead like structure called nucleosome. Nucleosome is a complex of DNA and histones. The histone core particle contains 8 histone molecules (H2a, H2b, H3 and H4 - dimer). The surface of the core particle is surrounded by a flat super helical strand of DNA that makes 1.75 turns. The adjacent nucleosomes are connected by linker DNA. A single histone H1 binds to the linker DNA. Histone core helps in the tight coiling of DNA.
During interphase certain regions of the chromatin stain darker with fuelgen. Such regions are called heterochromatins. The other regions which have lighter stains are called as euchromatin. This phenomenon is known as heteropycnosis or differential staining. Heterochromatin is associated with tight folding and coiling of the chromosome fibre. It contains relatively few genes in relation to its length. The DNA of heterochromatin is genetically inert and does not transcribe mRNA for protein synthesis. It contains genes for rRNA, 5SRNA, and tRNA. Part of heterochromain is also called as satellite DNA or repetitive DNA. Heterochromatin forms centromeric region of chromosome. It prevents mutation and crossing over. It is responsible for homologous chromosome pairing during meiosis. Euchromatin region is non-condensed region. Euchromatin form unique gene region of the genome. It is actively participate in transcription.
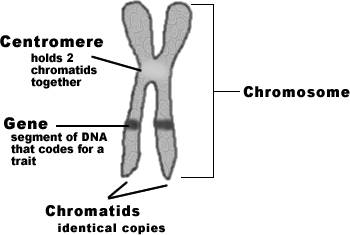
During mitotic phase, chromatin fibre condensed to form chromosomes. Chromosomes are self-reproducing thread like structures located inside the nucleus. They are called chromososme ( Chromo = color soma=body) because they are easily stained with dyes. The number of chromosomes varies from species to species but the number remains constant within species. The lowest number of chromosome is two in Ascaris megalocephala and highest is 1700 in radiolarian (protozoa). 46 chromosomes (23 pairs) present in human. A typical chromosome has an elongated cylindrical body with two arms namely P and Q arms. It consists of centromere, telomere, chromatids and secondary constriction (NOR). Chromosomes control heredity. They control metabolism of an organism. Changes in the position, number and the structure of chromosomes lead to the formation of new species.
FUNCTIONS OF NUCLEUS:
Metabolism: Nucleus controls manority of the activities of cells. It is regulatory organelle in cell metabolism.
Heredity: Since the nucleus contains DNA molecules in its chromosomes, it plays a significant role in heredity.
Differentiation: It controls cell differentiation during the embryonic development.
Exchange of materials: Nuclear membrane is concerned with the exchange of materials between the cytoplasm and nucleoplasm.
Support: Nuclear membrane provides a surface for the attachment of structural elements of the cytoplasm such microtubules and microfilaments.
RNA Synthesis: The syntheis of RNA occurs with in nucleus. From these RNAs ribosome, tRNA and various proteins are formed.
NULCEAR PORE COMPLEX:
The nuclear pore complexes are the only channels through which small polar molecules, ions, and macromolecules (proteins and RNAs) are able to travel between the nucleus and the cytoplasm. The nuclear pore complex is an extremely large structure with a diameter of about 120 nm and an estimated molecular mass of approximately 125 million daltons—about 30 times the size of a ribosome. In vertebrates, the nuclear pore complex is composed of 50 to 100 different proteins. By controlling the traffic of molecules between the nucleus and cytoplasm, the nuclear pore complex plays a fundamental role in the physiology of all eukaryotic cells. RNAs that are synthesized in the nucleus must be efficiently exported to the cytoplasm, where they function in protein synthesis. Conversely, proteins required for nuclear functions (e.g., transcription factors) must be transported into the nucleus from their sites of synthesis in the cytoplasm. In addition, many proteins shuttle continuously between the nucleus and the cytoplasm. The regulated traffic of proteins and RNAs through the nuclear pore complex thus determines the composition of the nucleus and plays a key role in gene expression.
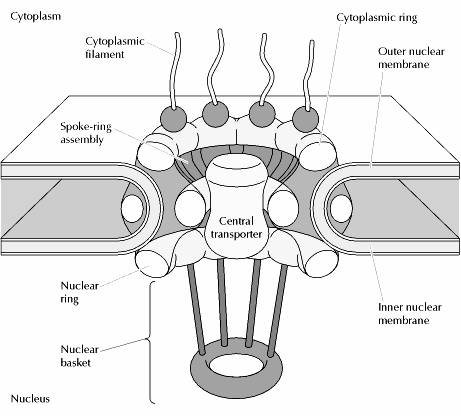
Visualization of nuclear pore complexes by electron microscopy reveals a structure with eight fold symmetry organized around a large central channel, which is the route through which proteins and RNAs cross the nuclear envelope. Detailed structural studies, including computer-based nucl analysis, have led to the development of three-dimensional models of the nuclear pore complex. These studies indicate that the nuclear pore complex consists of an assembly of eight spokes arranged around a central channel. The spokes are connected to rings at the nuclear and cytoplasmic surfaces, and the spoke-ring assembly is anchored within the nuclear envelope at sites of fusion between the inner and outer nuclear membranes. Protein filaments extend from both the cytoplasmic and nuclear rings, forming a distinct basketlike structure on the nuclear side. The central channel is approximately 40 nm in diameter, which is wide enough to accommodate the largest particles able to cross the nuclear envelope. It contains a structure called the central transporter, through which the active transport of macromolecules is thought to occur.
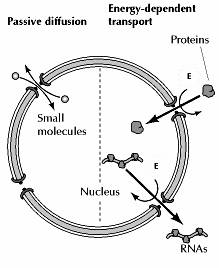
Depending on their size, molecules can travel through the nuclear pore complex by one of two different mechanisms. Small molecules and some proteins with molecular mass less than approximately 50 kd pass freely across the nuclear envelope in either direction: cytoplasm to nucleus or nucleus to cytoplasm. These molecules diffuse passively through open aqueous channels, estimated to have diameters of approximately 9 nm, in the nuclear pore complex. Most proteins and RNAs, however, are unable to pass through these open channels. Instead, these macromolecules pass through the nuclear pore complex by an active process in which appropriate proteins and RNAs are recognized and selectively transported in only one direction (nucleus to cytoplasm or cytoplasm to nucleus). The traffic of these molecules occurs through regulated channels in the nuclear pore complex that, in response to appropriate signals, can open to a diameter of more than 25 nm—a size sufficient to accommodate large ribonucleoprotein complexes, such as ribosomal subunits. It is through these regulated channels that nuclear proteins are selectively imported from the cytoplasm to the nucleus while RNAs are exported from the nucleus to the cytoplasm.
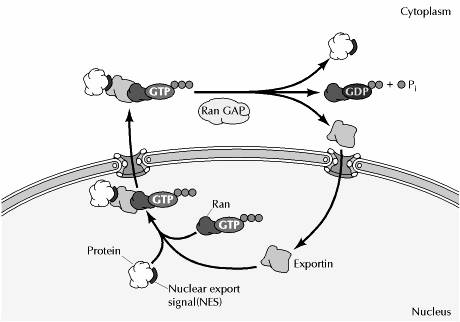
Nuclear import:
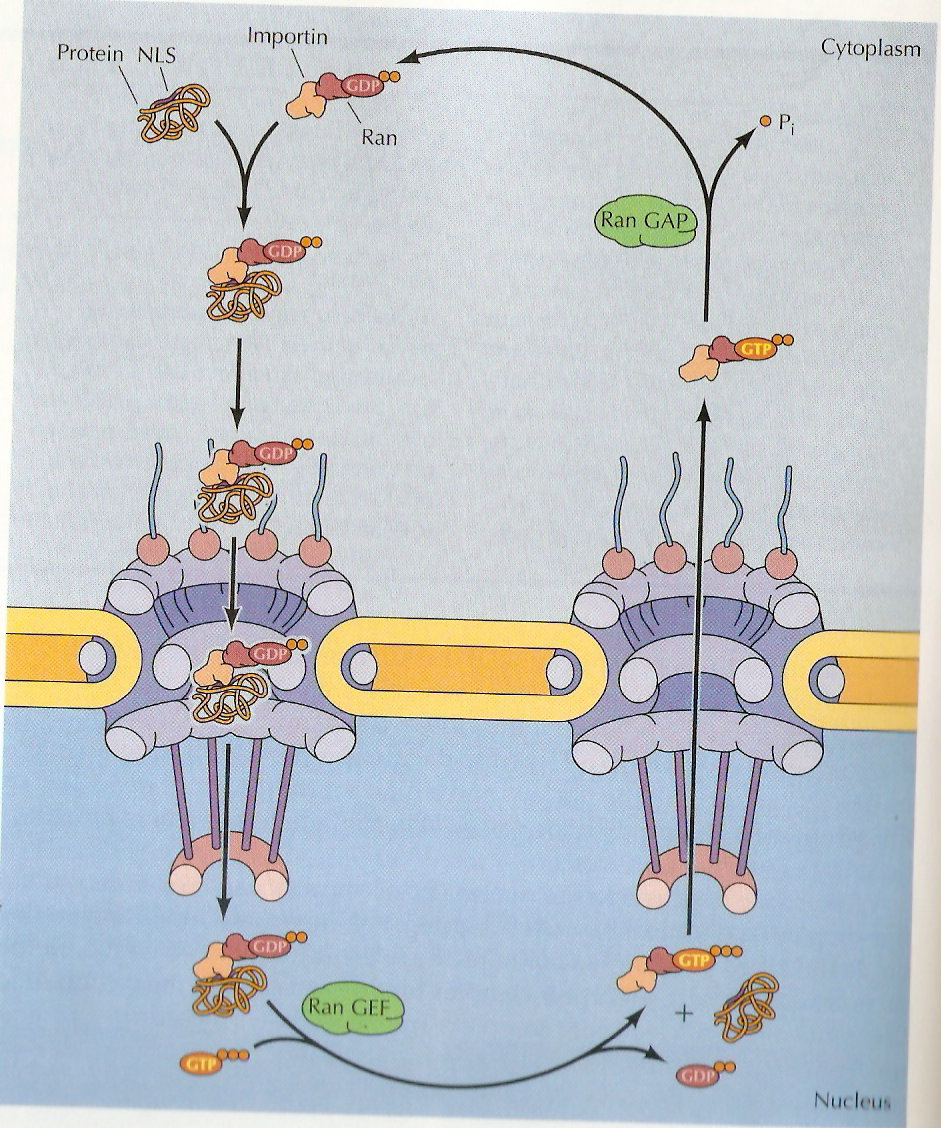
Proteins are imported through the nuclear pore complex in a five step cycle. In the first step, the protein with a nuclear localization signal sequence (NLS) is recognized by an importin complexed to the small GTP-binding protein, Ran (Ras releated proteins). In step two, the protein with nuclear localization signal-importin-rad/GDP complex binds to a specific nuclear pore protein in the cytoplasmic filaments. In the step three, the complex is translocated through the nuclear pore by sequential binding to pore proteins. In step four, the activity of the guanine nucleotide exchange factor (GEF) in the nucleus exchanges the GDP on Ran for GTP, altering the configuration of the complex so that the protein is released. In step five, this importin-Ran/GTP complex is re-exported through the nuclear pore and the GTP-ase activating protein (GAP) in the cytoplasm hydrolyses the GTP on Ran to Ran-GDP. Now Ran-GDP complex ready for next round of import.
Nuclear export:
Proteins are targeted for export from the nucleus by specific amino acid sequences, called nuclear export signals. Like nuclear localization signals, nuclear export signals are recognized by receptors within the nucleus that direct protein transport through the nuclear pore complex to the cytoplasm. Interestingly, the nuclear export receptors (called exportins) are related to importin. Like importin, the exportins bind to Ran, which is required for nuclear export as well as for nuclear import. Strikingly, however, Ran/GTP promotes the formation of stable complexes between exportins and their target proteins, whereas it dissociates the complexes between importins and their targets. This effect of Ran/GTP binding on exportins dictates the movement of proteins containing nuclear export signals from the nucleus to the cytoplasm. Thus, exportins form stable complexes with their target proteins in association with Ran/GTP within the nucleus. Following transport to the cytosolic side of the nuclear envelope, GTP hydrolysis leads to dissociation of the target protein, which is released into the cytoplasm.
Nucleolus:
The most prominent substructure within the nucleus is the nucleolus, which is the site of rRNA transcription and processing, and of ribosome assembly. Cells require large numbers of ribosomes to meet their needs for protein synthesis. Actively growing mammalian cells, for example, contain 5 million to 10 million ribosomes that must be synthesized each time the cell divides. The nucleolus is a ribosome production factory, designed to fulfill the need for large-scale production of rRNAs and assembly of the ribosomal subunits.
Nucleolus usually present in interphase cells. It is absent during division pahse. It was first discovered by Fontana in 1781. Nucleolus is absent from lower organisms like bacteria, yeast, mammalian erythrocytes, reticulocytes etc. In all other nuclear cells, nucleolus present.
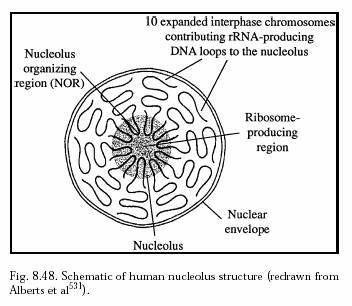
The number of nucleoli depends upon the number of sets of chromosomes. Usually one nucleolus is present for each set of chromosome. A haploid cell contains only one nucleolus. However, amphibian oocyte contains 600 to 1200 nucleoli. The nucleolus is located in the nucleolar organizer region (NOR) of the nucleolar chromosome. The size of the nucleolus depends upon the synthetic activity of the cell. The nucleoli are small or absent in cells exhibiting little protein synthesis.
Nucleolus structure:
The nucleolus is surrounded by a thick covering called perinucleolar chromatin. It may be continuous or with holes. The perinucleolar chromatin projects in some places into the nucleolus to form intranucleolar chromatin. Chromatins serves as the template for the synthesis of rRNA. The interior of the nucleolus is filled with a proteineous ground substance called matrix or pars amorpha. The matrix also contains fibrils and granules. The firbrils contain rRNA. The granules contain protein and RNA. The enzymes present in nucleolus include acid phosphatase, nucleoside phosphorylase, RNA methylase etc.
Nucleoli are classified into three types based on the distribution of granules. Homogenous nucleolus – The granules are uniformly distributed throughout the nucleus. Heterogenous nucleolus – The granules are occurring in groups. Ring nucleolus – The granules are arranged along the periphery of the nucleolus in the form of ring.
Nucleolus function:
1. Transcription and Processing of rRNA
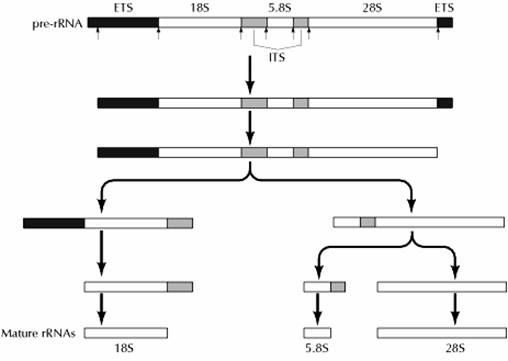
The primary transcript of the rRNA genes is the large 45S pre-rRNA, which contains the 18S, 5.8S, and 28S rRNAs as well as transcribed spacer regions (Fig 7.18). External transcribed spacers are present at both the 5′ and 3′ ends of the pre-rRNAs, and two internal transcribed spacers lie between the 18S, 5.8S, and 28S rRNA sequences. The initial processing step is a cleavage within the external transcribed spacer near the 5′ end of the pre-rRNA, which takes place during the early stages of transcription. This cleavage requires the U3 small nucleolar RNP that remains attached to the 5′ end of the pre-rRNA, forming the characteristic knobs. Once transcription is complete, the external transcribed spacer at the 3′ end of the molecule is removed. In human cells, this step is followed by a cleavage at the 5′ end of the 5.8S region, yielding separate precursors to the 18S and 5.8S + 28S rRNAs. Additional cleavages then result in formation of the mature rRNAs. Processing follows a similar pattern in other species, although there are differences in the order of some of the cleavages.
In addition to cleavage, the processing of pre-rRNA involves a substantial amount of base modification resulting both from the addition of methyl groups to specific bases and ribose residues and from the conversion of uridine to pseudouridine. In animal cells, pre-rRNA processing involves the methylation of approximately a hundred ribose residues and ten bases, in addition to the formation of about a hundred pseudouridines. Most of these modifications occur during or shortly after synthesis of the pre-rRNA, although a few take place at later stages of pre-rRNA processing.
Each nucleolar organizing region contains a cluster of tandemly repeated rRNA genes that are separated from each other by nontranscribed spacer DNA. These genes are very actively transcribed by RNA polymerase I, allowing their transcription to be readily visualized by electron microscopy. In such electron micrographs, each of the tandemly arrayed rRNA genes is surrounded by densely packed growing RNA chains, forming a structure that looks like a Christmas tree. The high density of growing RNA chains reflects that of RNA polymerase molecules, which are present at a maximal density of approximately one polymerase per hundred base pairs of template DNA.
2. Ribosome Assembly:
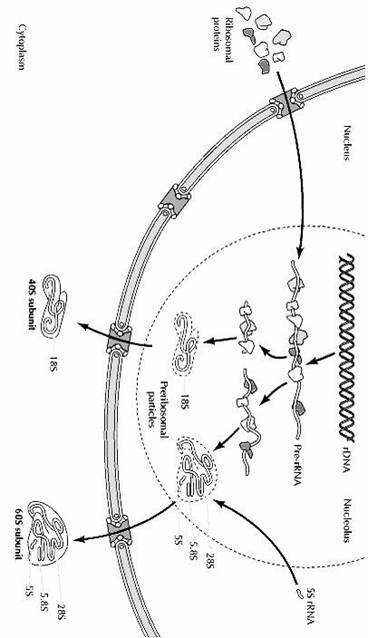
The formation of ribosomes involves the assembly of the ribosomal precursor RNA with both ribosomal proteins and 5S rRNA (Fige 7.20). The genes that encode ribosomal proteins are transcribed outside of the nucleolus by RNA polymerase II, yielding mRNAs that are translated on cytoplasmic ribosomes. The ribosomal proteins are then transported from the cytoplasm to the nucleolus, where they are assembled with rRNAs to form preribosomal particles. Although the genes for 5S rRNA are also transcribed outside of the nucleolus, in this case by RNA polymerase III, 5S rRNAs similarly are assembled into preribosomal particles within the nucleolus.
The association of ribosomal proteins with rRNA begins while the pre-rRNA is still being synthesized, and more than half of the ribosomal proteins are complexed with the pre-rRNA prior to its cleavage. The remaining ribosomal proteins and the 5S rRNA are incorporated into preribosomal particles as cleavage of the pre-rRNA proceeds. The smaller ribosomal subunit, which contains only the 18S rRNA, matures more rapidly than the larger subunit, which contains 28S, 5.8S, and 5S rRNAs. Consequently, most of the preribosomal particles in the nucleolus represent precursors to the large subunit. The final stages of ribosome maturation follow the export of preribosomal particles to the cytoplasm, forming the active 40S and 60S subunits of eukaryotic ribosomes.
Heterogenous nuclear RNA (hnRNA)
Heterogeneous nuclear RNA (hnRNA) is also called pre-mRNA. It's an incompletely-processed single strand of ribonucleic acid (RNA), and when it's completely processed it becomes mature mRNA.
Precursor mRNA (pre-mRNA) is an immature single strand of messenger ribonucleic acid (mRNA). pre-mRNA is synthesized from a DNA template in the cell nucleus by transcription. pre-mRNA comprises the bulk of heterogeneous nuclear RNA (hnRNA). The term hnRNA is often used as a synonym for pre-mRNA, although strictly speaking hnRNA may include nuclear RNA transcripts that do not end up as cytoplasmic mRNA.
Once pre-mRNA has been completely processed, it is termed "mature messenger RNA", "mature mRNA", or simply "mRNA".
hnRNA is processed by spliceosomes, small organelles in the nucleus that are composed of protein and RNA.Eukaryotic Pre-mRNA exists only briefly before it is fully processed into mRNA. Pre-mRNAs include two different types of segments, exons and introns. Exons are segments that are retained in the final mRNA, while introns are removed in a process called splicing, which is performed by the spliceosome (except for self-splicing introns).
In Prokaryotes, the splicing is done either autocatalytically or through endolytic cleavage. Autocatalytic cleavages, in which no proteins are involved, are usually reserved for sections that code for rRNA while endolytic cleavage corresponds to tRNA precursors.
Additional processing steps attach modifications to the 5' and 3' ends of the prokaryotic pre-mRNA. These include a 5' cap of 7-methylguanosine and a poly-A tail. Additionally, Eukaryotic pre-mRNAs have their introns spliced out by spliceosomes made up of Small Nuclear Ribo-Nucleo Proteins.
When a pre-mRNA strand has been properly processed to an mRNA sequence, it is exported out of the nucleus and eventually translated into a protein – a process accomplished in conjunction with ribosomes.