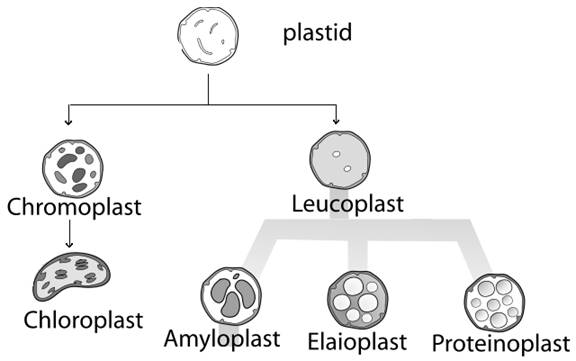
PLASTIDS
Plastids are large cytoplasmic organelles. Plastids are major organelles found in the cells of plants and algae. Plastids are the site of manufacture and storage of important chemical compounds used by the cell. Plastids often contain pigments used in photosynthesis, and the types of pigments present can change or determine the cell's colour. The term plastid was derived from the Greek word plastikas meaning formed or moulded. This term was coined by Schimper in 1885.
In plants, plastids may differentiate into several forms, depending upon which function they need to play in the cell. The plastids are broadly classified into two main types namely chromoplasts and leucoplasts.

Chromoplast:
These are colored plastids (chromo=color; plast=living). They contain various pigments. They synthesize food materials by photosynthesis. They contain yellow, orange and or red pigments. Chromoplasts are found commonly in flowers and fruits. Chromoplasts also divided into three types based on their color namely chloroplast, phaeoplast and rhodoplast.
Chloroplast: It is in green color. It contains chlorophyll pigments. It is found in higher plants and green algae.
Phaeoplast: It is dark brown in color. It contains fucoxanthin pigments. It is found in brown algae, diatoms and dinoflagelates.
Rhodoplast: It is red in color. It contains phycoerythrin. It is found in red algae.
Leucoplasts:
They are non-pigmented plastids (Leuco=white; plast=living). Their main function is to store food materials. They do not involve in synthetic activities. The leucoplasts are subdivided into three types namely amlyoplast, elaioplast and proteinoplast.
Amlyloplast: It stores starch and found in tubers, cotyledons and endosperm
Elaioplast: it stores oil and found in the epidermal cells.
Proteinoplast: It stores protein and found in seeds and nuts.
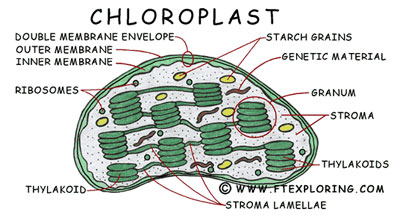
CHLOROPLASTS
Chloroplasts are organelles found in plant cells and other eukaryotic organisms that conduct photosynthesis. The word chloroplast is derived from the Greek words chloros, which means green, and plast, which means form or entity. Chloroplasts are members of a class of organelles known as plastids.
Shape:
Chloroplast varies in shape. They are spheroid or ovoid or discoid in higher plants. They are cup-shaped in chlamydomonas and spirally coiled in spirogyra.
Size:
The size of the plastids varies from species to species. But the size remains constant for a given cell type. In higher plants, it is 4-5microns in length and 1-3microns in thickness. Generally chroloplasts of plants growing in shady places are larger in size.
Number:
The number of chloroplasts varies from plant to plant, but it remains constant for a given plant. In higher plants there are 20 to 40 chloroplasts per cell or upto 1000 chloroplasts.
Structure:
Plant chloroplasts are large organelles (5 to 10 μm long) that, like mitochondria, are bounded by a double membrane called the chloroplast envelope. In addition to the inner and outer membranes of the envelope, chloroplasts have a third internal membrane system, called the thylakoid membrane. The thylakoid membrane forms a network of flattened discs called thylakoids, which are frequently arranged in stacks called grana. Grana are interconnected by branching membraneous tubules called frets (stromal lamellae). Because of this three-membrane structure, the internal organization of chloroplasts is more complex than that of mitochondria. In particular, their three membranes divide chloroplasts into three distinct internal compartments: (1) the intermembrane space between the two membranes of the chloroplast envelope; (2) the stroma, which lies inside the envelope but outside the thylakoid membrane; and (3) the thylakoid lumen.
A thylakoid has a flattened disk shape. Inside it is an empty area called the thylakoid space or lumen. Photosynthesis takes place on the thylakoid membrane; as in mitochondrial oxidative phosphorylation, it involves the coupling of cross-membrane fluxes with biosynthesis via the dissipation of a proton electrochemical gradient. Embedded in the thylakoid membrane are antenna complexes, each of which consists of the light-absorbing pigments, including chlorophyll and carotenoids, as well as proteins that bind the pigments. These complexes are called as quantosomes. This complex both increases the surface area for light capture, and allows capture of photons with a wider range of wavelengths. The energy of the incident photons is absorbed by the pigments and funneled to the reaction centre of this complex through resonance energy transfer. Two chlorophyll molecules are then ionised, producing an excited electron, which then passes onto the photochemical reaction centre.
Chloroplast contains proteins, lipids, carbohydrates, DNA, RNA, carotenoids, chlorophyll and minerals. Composition of these chemical were indicated in the following table:
|
S. No |
Chemical |
Percentage |
|
1. |
Proteins |
35 – 55% |
|
2. |
Lipids |
20 – 30% |
|
3. |
Carbohydrates |
Variable |
|
4. |
Chlorophyll |
9% |
|
5. |
Carotenoids |
4.5% |
|
6. |
RNA |
3 – 4% |
|
7. |
DNA |
0.5% |
|
8. |
Minerals |
0.2% |
Photosynthetic Pigments:
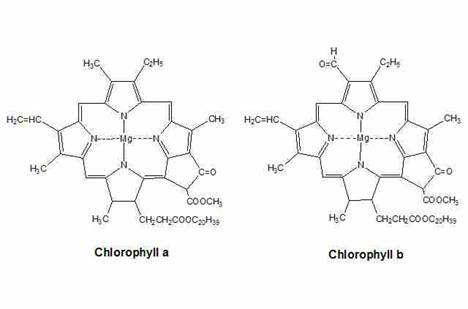
Pigments carrying out photosynthesis are called as photosynthetic pigments. Chloroplast also contains photosynthetic pigments. They are chlorophylls, carotenoids and phycobilins. Chlorophyll a absorbs well at a wavelength of about 400-450 nm and at 650-700 nm; chlorophyll b at 450-500 nm and at 600-650 nm. Xanthophyll absorbs well at 400-530 nm. However, none of the pigments absorbs well in the green-yellow region, which is responsible for the abundant green we see in nature.
Chlorophylls are greenish pigments which contain a porphyrin ring. This is a stable ring-shaped molecule around which electrons are free to migrate. Because the electrons move freely, the ring has the potential to gain or lose electrons easily, and thus the potential to provide energized electrons to other molecules. This is the fundamental process by which chlorophyll "captures" the energy of sunlight.
There are several kinds of chlorophyll, the most important being chlorophyll "a". This is the molecule which makes photosynthesis possible, by passing its energized electrons on to molecules which will manufacture sugars. All plants, algae, and cyanobacteria which photosynthesize contain chlorophyll "a". A second kind of chlorophyll is chlorophyll "b", which occurs only in "green algae" and in the plants. A third form of chlorophyll which is common is (not surprisingly) called chlorophyll "c", and is found only in the photosynthetic members of the Chromista as well as the dinoflagellates. The differences between the chlorophylls of these major groups was one of the first clues that they were not as closely related as previously thought.
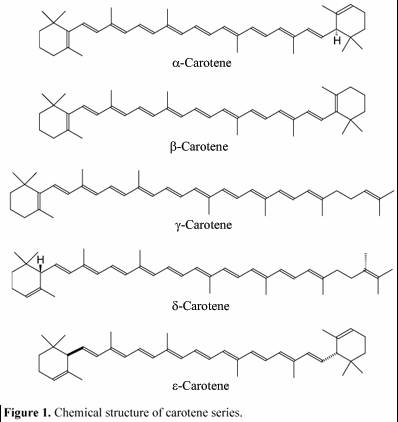
Carotenoids are usually red, orange, or yellow pigments, and include the familiar compound carotene, which gives carrots their color. These compounds are composed of two small six-carbon rings connected by a "chain" of carbon atoms. As a result, they do not dissolve in water, and must be attached to membranes within the cell. Carotenoids cannot transfer sunlight energy directly to the photosynthetic pathway, but must pass their absorbed energy to chlorophyll. For this reason, they are called accessory pigments. One very visible accessory pigment is fucoxanthin the brown pigment which colors kelps and other brown algae as well as the diatoms.
Phycobilins are water-soluble pigments, and are therefore found in the cytoplasm, or in the stroma of the chloroplast. They occur only in Cyanobacteria and Rhodophyta. The picture at the right shows the two classes of phycobilins which may be extracted from these "algae". The vial on the left contains the bluish pigment phycocyanin, which gives the Cyanobacteria their name. The vial on the right contains the reddish pigment phycoerythrin, which gives the red algae their common name.
Phycobilins are not only useful to the organisms which use them for soaking up light energy; they have also found use as research tools. Both pycocyanin and phycoerythrin fluoresce at a particular wavelength. That is, when they are exposed to strong light, they absorb the light energy, and release it by emitting light of a very narrow range of wavelengths. The light produced by this fluorescence is so distinctive and reliable, that phycobilins may be used as chemical "tags". The pigments are chemically bonded to antibodies, which are then put into a solution of cells. When the solution is sprayed as a stream of fine droplets past a laser and computer sensor, a machine can identify whether the cells in the droplets have been "tagged" by the antibodies. This has found extensive use in cancer research, for "tagging" tumor cells.
Chloroplast Transport:
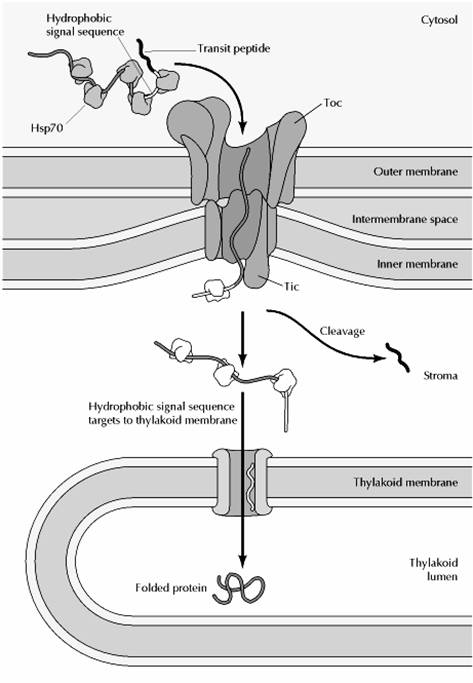
Most chloroplast proteins are synthesized on free ribosomes in the cytosol and targeted for import to chloroplasts by amino-terminal transit peptides. Proteins incorporated into the thylakoid lumen are first imported into the chloroplast stroma and then targeted for transport across the thylakoid membrane by a second hydrophobic signal sequence.
FUNCTIONS OF CHLOROPLAST
Chloroplast has the following functions:
1. Protein Synthesis:
As chloroplast contains its own DNA and ribososmes, it synthesizes certain amount of proteins for its metabolism.
2. Startch Storage:
Chloroplasts temporarily store starch during the day time in the pyrenoid which is a starch forming organelle. At night the starch is transferred to regions of growth and storage.
4. Oxygen Supply:
Chloroplasts utilize carbondioxide for photosynthesis and release oxygen. This oxygen is used by all animals and plants for respiration. Oxygen release occurs during photophosphorylation process of light reactions. During light reaction, water lytic centre or oxygen evolving centre, release oxygen by breaking water molecules in the presence of light by photosystem-II.
5. Photosynthesis:
Photosynthesis (from the Greek photo, "light," and synthesis, "putting together.") is a process that converts carbon dioxide into organic compounds, especially sugars, using the energy from sunlight. Photosynthesis occurs in plants, algae, and many species of Bacteria, but not in Archaea. Photosynthetic organisms are called photoautotrophs, since it allows them to create their own food. In plants, algae and cyanobacteria photosynthesis uses carbon dioxide and water, releasing oxygen as a waste product. Photosynthesis is vital for life on Earth.
2n CO2 + 2n H2O + photons → 2(CH2O)n + 2n O2
Carbondioxide + water + light energy → carbohydrate + oxygen
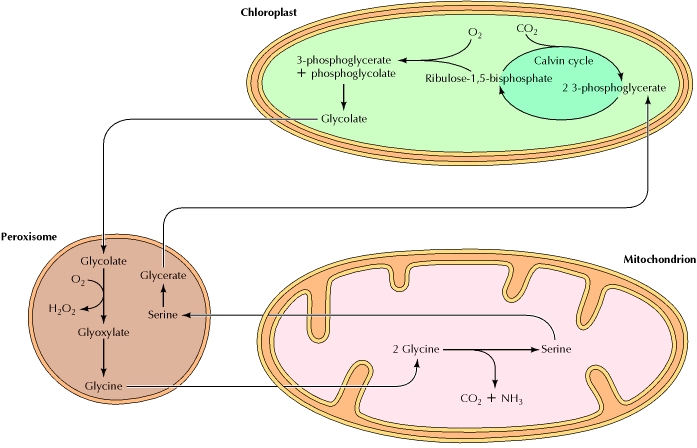
6. Photorespiration:
Photorespiration is a process when Ribulose bisphosphate carboxylase / oxygenase (RUBisco) fix oxygen instead of carbondioxide. Photorespiration can occur when carbon dioxide levels are low. Photorespiration produces no ATP (energy for cells) and leads to a net loss of carbon and nitrogen (as ammonia) which slows the growth of plants. Photorespiration is said to be an evolutionary relic. Photorespiration lowers the efficiency of photosynthesis by removing carbon molecules from the Calvin Cycle.
In photorespiration, the enzyme involved (ribulose bisphosphate carboxylase or rubisco) sometimes catalyzes the addition of O2 instead of CO2, producing one molecule of 3-phosphoglycerate and one molecule of phosphoglycolate (two carbons). This is a side reaction, and phosphoglycolate is not a useful metabolite. It is first converted to glycolate and then transferred to peroxisomes, where it is oxidized and converted to glycine. Glycine is then transferred to mitochondria, where two molecules of glycine are converted to one molecule of serine, with the loss of CO2 and NH3. The serine is then returned to peroxisomes, where it is converted to glycerate. Finally, the glycerate is transferred back to chloroplasts, where it reenters the Calvin cycle. Photorespiration does not appear to be beneficial for the plant, since it is essentially the reverse of photosynthesis—O2 is consumed and CO2 is released without any gain of ATP. However, the occasional utilization of O2 in place of CO2 appears to be an inherent property of rubisco, so photorespiration is a general accompaniment of photosynthesis.
PHOTOSYNTHESIS
Photosyntesis takes place in the chloroplasts in green plants and eukaryotic algae and within photosynthetic membranes in prokaryotes. The process of photosynthesis takes place in two steps. They are Light reaction an Dark reaction. In the light reactions, energy from sunlight drives the synthesis of ATP and NADPH, coupled to the formation of O2 from H2O. In the dark reactions, so named because they do not require sunlight, the ATP and NADPH produced by the light reactions drive glucose synthesis. In eukaryotic cells, both the light and dark reactions of photosynthesis occur within chloroplasts—the light reactions in the thylakoid membrane and the dark reactions within the stroma.
LIGHT REACTION:
In the light reactions, one molecule of the pigment chlorophyll absorbs one photon and loses one electron. It occurs only in the presence of light and hence the name light reaction. During light reaction light energy (photon) is converted into chemical energy (ATP). There are twp steps in light reactions, namely photolysis and photophosphorylation. In photolysis, water is split into electrons, hydrogen and oxygen. In photophosphorylation, ATP is synthesized. Photophosphorylation occurs in two ways namely Non-cyclic photophosphorylation and cyclic photophosphorylation.
Non-cyclic photophosphorylation:
The pathway of electron flow starts at photosystem II, which is homologous to the photosynthetic reaction center of R. viridis already described. However, at photosystem II the energy derived from absorption of photons is used to split water molecules to molecular oxygen and protons. This reaction takes place within the thylakoid lumen, so the release of protons from H2O establishes a proton gradient across the thylakoid membrane. The high-energy electrons derived from this process are transferred through a series of carriers to plastoquinone, a lipid-soluble carrier similar to coenzyme Q (ubiquinone) of mitochondria. Plastoquinone carries electrons from photosystem II to the cytochrome bf complex, within which electrons are transferred to plastocyanin and additional protons are pumped into the thylakoid lumen. Electron transport through photosystem II is thus coupled to establishment of a proton gradient, which drives the chemiosmotic synthesis of ATP.
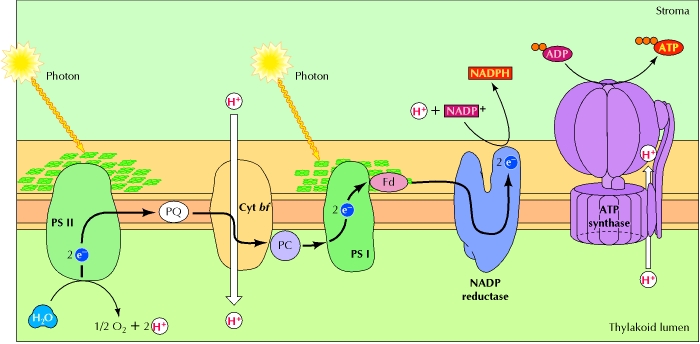
From photosystem II, electrons are carried by plastocyanin (a peripheral membrane protein) to photosystem I, where the absorption of additional photons again generates high-energy electrons. Photosystem I, however, does not act as a proton pump; instead, it uses these high-energy electrons to reduce NADP+ to NADPH. The reaction center chlorophyll of photosystem I transfers its excited electrons through a series of carriers to ferrodoxin, a small protein on the stromal side of the thylakoid membrane. The enzyme NADP reductase then transfers electrons from ferrodoxin to NADP+, generating NADPH. The passage of electrons through photosystems I and II thus generates both ATP and NADPH, which are used by the Calvin cycle enzymes in the chloroplast stroma to convert CO2 to carbohydrates. Electrons are shuttled through an electron transport chain, like “Z” letter so the process called as Z-scheme, that initially functions to generate a chemiosmotic potential across the membrane.
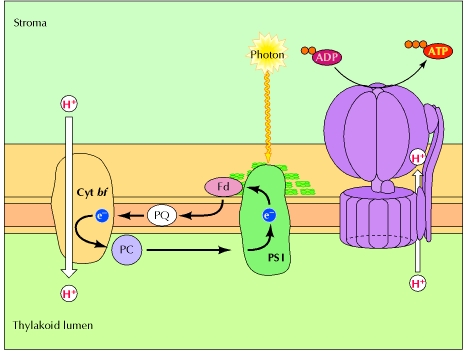
Cyclic Photophosphorylation:
A second electron transport pathway, called cyclic electron flow, produces ATP without the synthesis of NADPH, thereby supplying additional ATP for other metabolic processes. In cyclic electron flow, light energy harvested at photosystem I is used for ATP synthesis rather than NADPH synthesis. Instead of being transferred to NADP+, high-energy electrons from photosystem I are transferred to the cytochrome bf complex. Electron transfer through the cytochrome bf complex is then coupled, as in photosystem II, to the establishment of a proton gradient across the thylakoid membrane. Plastocyanin then returns these electrons to photosystem I in a lower energy state, completing a cycle of electron transport in which light energy harvested at photosystem I is used to pump protons at the cytochrome bf complex. Electron transfer from photosystem I can thus generate either ATP or NADPH, depending on the metabolic needs of the cell.
ATP Synthesis:
The ATP synthase of the thylakoid membrane is similar to the mitochondrial enzyme. However, the energy stored in the proton gradient across the thylakoid membrane, in contrast to the inner mitochondrial membrane, is almost entirely chemical in nature. This is because the thylakoid membrane, although impermeable to protons, differs from the inner mitochondrial membrane in being permeable to other ions, particularly Mg2+ and Cl-. The free passage of these ions neutralizes the voltage component of the proton gradient, so the energy derived from photosynthesis is conserved mainly as the difference in proton concentration (pH) across the thylakoid membrane. However, because the thylakoid lumen is a closed compartment, this difference in proton concentration can be quite large, corresponding to a differential of more than three pH units between the stroma and the thylakoid lumen. Because of the magnitude of this pH differential, the total free energy stored across the thylakoid membrane is similar to that stored across the inner mitochondrial membrane.
For each pair of electrons transported, two protons are transferred across the thylakoid membrane at photosystem II and two to four protons at the cytochrome bf complex. Since four protons are needed to drive the synthesis of one molecule of ATP, passage of each pair of electrons through photosystems I and II by noncyclic electron flow yields between 1 and 1.5 ATP molecules. Cyclic electron flow has a lower yield, corresponding to between 0.5 and 1 ATP molecules per pair of electrons.
DARK REACTIONS:
Dark reactions or Light independent reactions, converts corbondioxide into carbohydrate by using ATP and NADPH. This process also called as carbon fixation or carbon reaction since carbon is fixed during this reaction. It occurs in stroma of chloroplast. There are three pathways occurs in dark reaction, namely Calvin cyle, C 4 pathway and CAM pathway.
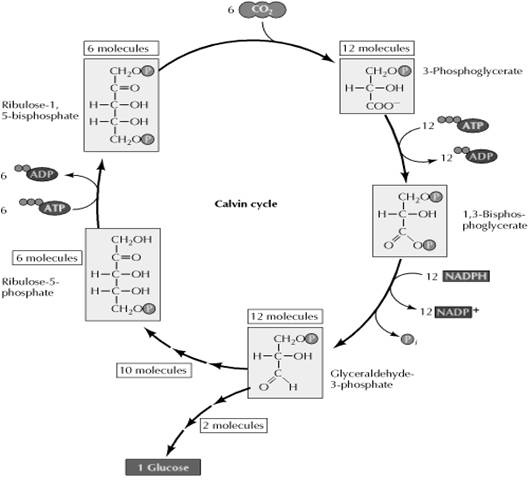
Calvin Cycle or C-3 pathway:
The path of carbon in dark reaction was worked out by Melvin Calvin. Hence it is called as Calvin cycle. As the three carbon compound 3-phosphoglycerate is the first stable product in this cycle, it is commonly called C-3 pathway. In this cycle, each molecule of CO2 is added to ribulose-1,5-bisphosphate to yield two molecules of 3-phosphoglycerate. Six molecules of CO2 thus lead to the formation of 12 molecules of 3-phosphoglycerate, which are converted to 12 molecules of glyceraldehyde-3-phosphate at the cost of 12 molecules each of ATP and NADPH. Two molecules of glyceraldehyde-3-phosphate are then used for synthesis of glucose and ten molecules continue in the Calvin cycle to form six molecules of ribulose-5-phosphate. The cycle is then completed by the use of six additional ATP molecules for the synthesis of ribulose-1,5-bisphosphate.
C-4 Pathway or Hatch-Slack pathway:
In hot and dry conditions, plants will close their stomata to prevent loss of water. Under these conditions, CO2 will decrease, and oxygen gas, produced by the light reactions of photosynthesis, will decrease in the stem, not leaves, causing an increase of photorespiration by the oxygenase activity of ribulose-1,5-bisphosphate carboxylase/oxygenase and decrease in carbon fixation. Some plants have evolved mechanisms to increase the CO2 concentration in the leaves under these conditions.
The name "C4" comes from the fact that the first product of CO2 fixation in these plants has four carbon atoms namely oxaloacetate. The C4 pathway was discovered by M. D. Hatch and C. R. Slack, in Australia, in 1966, so it is sometimes called the Hatch-Slack pathway. C4 plants chemically fix carbon dioxide in the cells of the mesophyll by adding it to the three-carbon molecule phosphoenolpyruvate (PEP), a reaction catalyzed by an enzyme called PEP carboxylase and which creates the four-carbon organic acid, oxaloacetic acid. Oxaloacetic acid or malate synthesized by this process is then translocated to specialized bundle sheath cells where the enzyme, rubisco, and other Calvin cycle enzymes are located, and where CO2 released by decarboxylation of the four-carbon acids is then fixed by rubisco activity to the three-carbon sugar 3-Phosphoglyceric acids. The physical separation of rubisco from the oxygen-generating light reactions reduces photorespiration and increases CO2 fixation and thus photosynthetic capacity of the leaf. C4 plants can produce more sugar than C3 plants in conditions of high light and temperature. Many important crop plants are C4 plants including maize, sorghum, sugarcane, and millet.
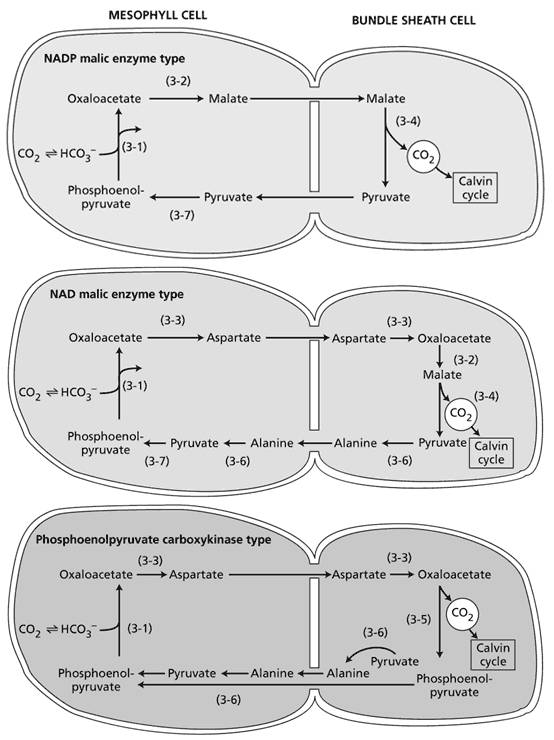
There are several variants of this pathway:
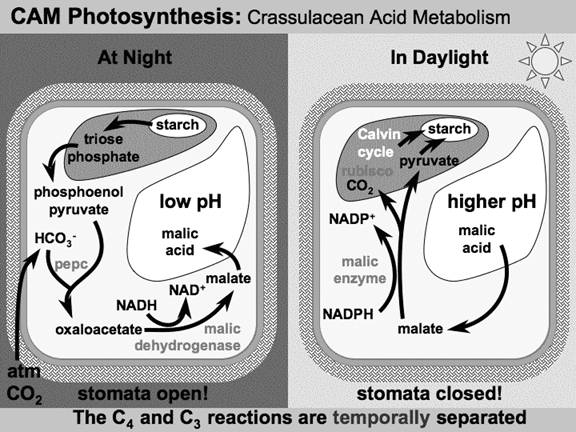
CAM or Crassulacean acid metabolism:
Xerophytes such as cacti and most succulents also use PEP carboxylase to capture carbon dioxide in a process called Crassulacean acid metabolism (CAM). In contrast to C4 metabolism, which physically separates the CO2 fixation to PEP from the Calvin cycle, CAM only temporally separates these two processes. CAM plants have a different leaf anatomy than C4 plants, and fix the CO2 at night, when their stomata are open. CAM plants store the CO2 mostly in the form of malic acid via carboxylation of phosphoenolpyruvate to oxaloacetate, which is then reduced to malate. Decarboxylation of malate during the day releases CO2 inside the leaves thus allowing carbon fixation to 3-phosphoglycerate by rubisco. The CAM pathway allows stomata to remain shut during the day; therefore it is especially common in plants adapted to arid conditions.
During the night
CAM plants open their stomata during the cooler and more humid night-time hours, permitting the uptake of carbon dioxide with minimal water loss.
The carbon dioxide is converted to soluble molecules, which can be readily stored by the plant at a sensible concentration.
The chemical pathway involves a three-carbon compound phosphoenolpyruvate (PEP), to which a CO2 molecule is added. This forms a new molecule, oxaloacetate, which in turn forms a malate. Oxaloacetate and malate are built around a skeleton of four carbons, hence the term C4. Malate can be readily stored by the plant in vacuoles within individual cells.
During the day
Malate can be broken down on demand, releasing a molecule of CO2 as it is converted to pyruvate. The pyruvate can be phosphorylated (i.e. have a phosphate group added by the "energy carrier" ATP) to regenerate the PEP with which the plant started, ready to be spurred into action the next night. But it is the release of CO2 that makes the cycle worth the plant's while. It is directed to the stroma of chloroplasts: the sites at which photosynthesis is most active. There, it is provided to RuBisCO in greater concentration, increasing the efficiency of the molecule, and therefore producing more sugars per unit photosynthesis.
MITOCHONDRIA
The word mitochondrion comes from the Greek mitos, thread + chondrion, granule. In cell biology, a mitochondrion (plural mitochondria) is a membrane-enclosed organelle found in most eukaryotic cells. Mitochondria are sometimes described as "cellular power plants" because they generate most of the cell's supply of adenosine triphosphate (ATP), used as a source of the chemical energy. The mitochondia were first discovered by Flemming and Kolliker. The term Mitochondria was introduced by Benda.
Number:
The number of mitochondria in a cell varies widely by organism and tissue type. Many cells have only a single mitochondrion, whereas others can contain several thousand mitochondria.
Size:
The size of the mitochondria is highly variable. These organelles range from 0.5–10 micrometers (μm) in diameter. The smallest mitochondrion is seen in yeast (1mMicron). The largest mitochondrion found in oocytes of amphibian (20 – 40 microns).
Distribution:
In most cells, the mitochondria are distributed uniformly throughout the cytoplasm. But in some cases they are aggregated around the nucleus. In Paramecium, they are located just beneath the surface of cells. In kidney tubules, they occur in the folds of basal regions near plasma membrane.
Shape:
The mitochondria may be filamentous or granular in shape. The shape of mitochondria may change from one to another depending upon the physiological conditions of the cell. They may be rod-shaped, club-shaped, ring-shaped, rounded or vesicular.
Structure:
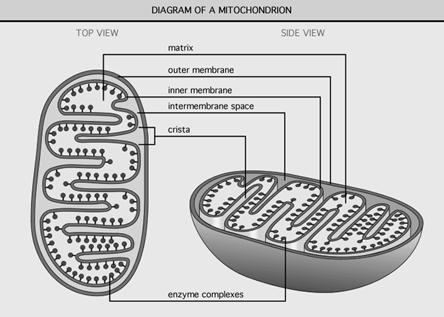
A mitochonstine contains outer and inner membranes composed of phospholipid bilayers and proteins. The two membranes, however, have different properties. Because of this double-membraned organization, there are five distinct compartments within the mitochondrion. There is the outer mitochondrial membrane, the intermembrane space (the space between the outer and inner membranes), the inner mitochondrial membrane, the cristae space (formed by infoldings of the inner membrane), and the matrix (space within the inner membrane).
The outer mitochondrial membrane, which encloses the entire organelle, has a protein-to-phospholipid ratio similar to that of the eukaryotic plasma membrane (about 1:1 by weight). It contains large numbers of integral proteins called porins. These porins form channels that allow molecules 5000 Daltons or less in molecular weight to freely diffuse from one side of the membrane to the other.
The intermembrane space is the space between the outer membrane and the inner membrane. Because the outer membrane is freely permeable to small molecules, the concentrations of small molecules such as ions and sugars in the intermembrane space are the same as the cytosol. However, as large proteins must have a specific signaling sequence to be transported across the outer membrane, the protein composition of this space is different than the protein composition of the cytosol. One protein that is localized to the intermembrane space in this way is cytochrome c.
It contains more than 100 different polypeptides, and has a very high protein-to-phospholipid ratio (more than 3:1 by weight, which is about 1 protein for 15 phospholipids). The inner membrane is home to around 1/5 of the total protein in a mitochondrion. In addition, the inner membrane is rich in an unusual phospholipid, cardiolipin. Cardiolipin contains four fatty acids rather than two and may help to make the inner membrane impermeable. Unlike the outer membrane, the inner membrane does not contain porins and is highly impermeable to all molecules. Almost all ions and molecules require special membrane transporters to enter or exit the matrix. Proteins are ferried into the matrix via the translocase of the inner membrane (TIM) complex or via Oxa1. In addition, there is a membrane potential across the inner membrane formed by the action of the enzymes of the electron transport chain.
The inner mitochondrial membrane is compartmentalized into numerous cristae, which expand the surface area of the inner mitochondrial membrane, enhancing its ability to produce ATP. These are not simple random folds but rather invaginations of the inner membrane, which can affect overall chemiosmotic function. In typical liver mitochondria, for example, the surface area, including cristae, is about five times that of the outer membrane. Mitochondria of cells that have greater demand for ATP, such as muscle cells, contain more cristae than typical liver mitochondria.These folds are studded with small round bodies known as F1 particles or oxysomes.
The matrix is the space enclosed by the inner membrane. It contains about 2/3 of the total protein in a mitochondrion. The matrix is important in the production of ATP with the aid of the ATP synthase contained in the inner membrane. The matrix contains a highly-concentrated mixture of hundreds of enzymes, special mitochondrial ribosomes, tRNA, and several copies of the mitochondrial DNA genome. Of the enzymes, the major functions include oxidation of pyruvate and fatty acids, and the citric acid cycle. Mitochondria have their own genetic material, and the machinery to manufacture their own RNAs and proteins. A published human mitochondrial DNA sequence revealed 16,569 base pairs encoding 37 total genes: 22 tRNA, 2 rRNA, and 13 peptide genes. The 13 mitochondrial peptides in humans are integrated into the inner mitochondrial membrane, along with proteins encoded by genes that reside in the host cell's nucleus.
The mitochondria are found to contain 65 to 75% protein, 25 to 30% lipid, 0.5% RNA and small amount of DNA. The lipid part of mitochondria is composed of 90% phospholipids, 5% cholesterol and 5% free fatty acids. Small amount of sulphur, iron copper, and some vitamins are present. There are more than 70 enzymes and co-enzymes in mitochondria.
FUNCTIONS OF MITOCHONDRIA:
Energy Supply:
A dominant role for the mitochondria is the production of ATP, as reflected by the large number of proteins in the inner membrane for this task. This is done by oxidizing the major products of glucose, pyruvate, and NADH, which are produced in the cytosol.
Cell respiration:
This process of cellular respiration, also known as aerobic respiration, is dependent on the presence of oxygen. When oxygen is limited, the glycolytic products will be metabolized by anaerobic respiration, a process that is independent of the mitochondria. The production of ATP from glucose has an approximately 13-fold higher yield during aerobic respiration compared to anaerobic respiration.
Heat Production:
Under certain conditions, protons can re-enter the mitochondrial matrix without contributing to ATP synthesis. This process is known as proton leak or mitochondrial uncoupling and is due to the facilitated diffusion of protons into the matrix. The process results in the unharnessed potential energy of the proton electrochemical gradient being released as heat. The process is mediated by a proton channel called thermogenin, or UCP1. Thermogenin is a 33kDa protein first discovered in 1973. Thermogenin is primarily found in brown adipose tissue, or brown fat, and is responsible for non-shivering thermogenesis. Brown adipose tissue is found in mammals, and is at its highest levels in early life and in hibernating animals. In humans, brown adipose tissue is present at birth and decreases with age.
Storage of calcium:
The concentrations of free calcium in the cell can regulate an array of reactions and is important for signal transduction in the cell. Mitochondria can transiently store calcium, a contributing process for the cell's homeostasis of calcium. In fact, their ability to rapidly take in calcium for later release makes them very good "cytosolic buffers" for calcium. The endoplasmic reticulum (ER) is the most significant storage site of calcium, and there is a significant interplay between the mitochondrion and ER with regard to calcium. The calcium is taken up into the matrix by a calcium uniporter on the inner mitochondrial membrane. It is primarily driven by the mitochondrial membrane potential. Release of this calcium back into the cell's interior can occur via a sodium-calcium exchange protein or via "calcium-induced-calcium-release" pathways. This can initiate calcium spikes or calcium waves with large changes in the membrane potential. These can activate a series of second messenger system proteins that can coordinate processes such as neurotransmitter release in nerve cells and release of hormones in endocrine cells.
Protein synthesis:
About 5 to 10% of proteins of mitochondria are synthesized by the mitochondrial DNA. Mitochondria synthesize subunits of ATPase, portion of reductase and three subunits of cytochrome oxidase.
Synthesis of steroid hormones:
The early step in the conversion of cholesterol to steroid hormones in the adrenal cortex, are catalyzed by mitochondrial enzymes.
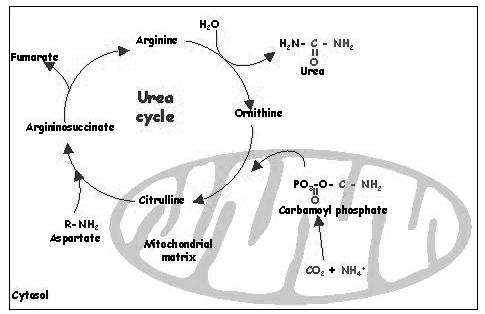
Urea cycle:
Urea cycle plays crucial role in ammonia detoxification. In this cycle, urea is synthesized. The first step of this cycle that is the conversion of ornithine to citrulline occurs in the mitochondria.
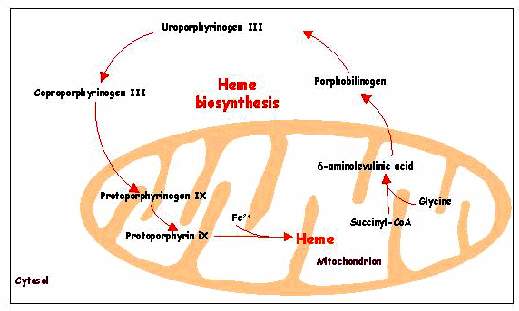
Heme Biosynthesis:
Heme, which is needed as a prosthetic group in several important proteins such as hemoglobin, myoglobin and cytochrome C, is partly synthesized in the mitochondrion. The condensation of succinyl-CoA and glycine to δ-aminolevulic acid is the key-step of the heme-synthesis and takes place in the mitochondrion. δ-Aminolevulic acid is then delivered into the cytosol where coproporphyrinogen III is formed after a series of reactions. This molecule later returns into the mitochondrion to be converted into heme.
Fatty acid oxidation:
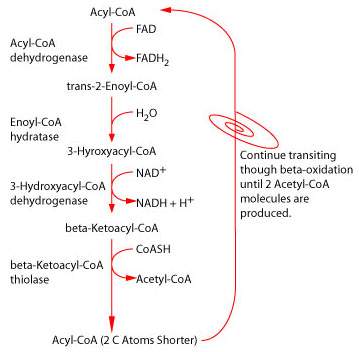
The carnitine-dependent transport of fatty acids and their β–oxidation is another important metabolic pathway located in the mitochondrion. Most of the fatty acids to be oxidized for energy production by intra-mitochondrial β-oxidation have to be transported from the cytosol into the mitochondrion. For transport, the fatty acids are first esterified with Coenzyme A (CoA) for “activation”, and are then coupled to carnitine to transverse the mitochondrial double membrane. All enzymes of the β-oxidation are mitochondrial enzymes. Acetyl-CoA, NADH, and FADH2, which are generated in each round of fatty acid oxidation, will later be channeled either into the citric acid cycle or directly into the respiratory chain to produce ATP.
CELLULAR RESPIRATION:
Cellular respiration, also known as 'oxidative metabolism', is one of the key ways a cell gains useful energy. It is the set of the metabolic reactions and processes that take place in organisms' cells to convert biochemical energy from nutrients into adenosine triphosphate (ATP), and then release waste products. The reactions involved in respiration are catabolic reactions that involve the oxidation of one molecule and the reduction of another.
Nutrients commonly used by animal and plant cells in respiration include glucose, amino acids and fatty acids, and a common oxidizing agent (electron acceptor) is molecular oxygen (O2). Bacteria and archaea can also be lithotrophs and these organisms may respire using a broad range of inorganic molecules as electron donors and acceptors, such as sulfur, metal ions, methane or hydrogen. Organisms that use oxygen as a final electron acceptor in respiration are described as aerobic, while those that do not are referred to as anaerobic.
The energy released in respiration is used to synthesize ATP to store this energy. The energy stored in ATP can then be used to drive processes requiring energy, including biosynthesis, locomotion or transportation of molecules across cell membranes.
Aerobic Respiration:
Aerobic respiration requires oxygen in order to generate energy (ATP). It is the preferred method of pyruvate breakdown from glycolysis and requires that pyruvate enter the mitochondrion in order to be fully oxidized by the Krebs cycle.
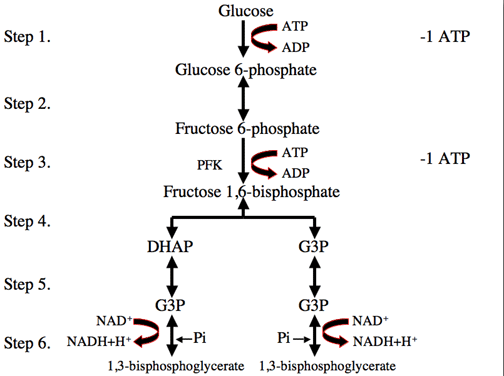
Glycolysis:
Glycolysis is a metabolic pathway that is found in the cytoplasm of cells in all living organisms and is anaerobic (that is, oxygen is not required). The process converts one molecule of glucose into two molecules of pyruvate, and makes energy in the form of two net molecules of ATP. Four molecules of ATP per glucose are actually produced; however, two are consumed for the preparatory phase. The initial phosphorylation of glucose is required to destabilize the molecule for cleavage into two triose sugars. During the pay-off phase of glycolysis, four phosphate groups are transferred to ADP by substrate-level phosphorylation to make four ATP, and two NADH are produced when the triose sugars are oxidized.
Oxidative decarboxylation of Pyruvate:
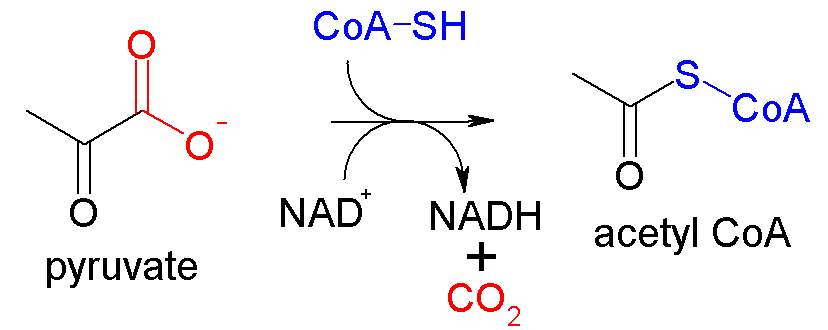
The pyruvate is oxidized to acetyl-CoA and CO2 by the Pyruvate dehydrogenase complex, a cluster of enzymes—multiple copies of each of three enzymes—located in the mitochondria of eukaryotic cells and in the cytosol of prokaryotes. In the process one molecule of NADH is formed per pyruvate oxidized, and 3 moles of ATP are formed for each mole of pyruvate. This step is also known as the link reaction, as it links glycolysis and the Krebs cycle.
Citric acid cycle:
This is also called the Krebs cycle or the tricarboxylic acid cycle. When oxygen is present, acetyl-CoA is produced from the pyruvate molecules created from glycolysis. Once acetyl-CoA is formed, two processes can occur, aerobic or anaerobic respiration. When oxygen is present, the mitochondria will undergo aerobic respiration which leads to the Krebs cycle. However, if oxygen is not present, fermentation of the pyruvate molecule will occur. In the presence of oxygen, when acetyl-CoA is produced, the molecule then enters the citric acid cycle (Krebs cycle) inside the mitochondrial matrix, and gets oxidized to CO2 while at the same time reducing NAD to NADH. NADH can be used by the electron transport chain to create further ATP as part of oxidative phosphorylation. To fully oxidize the equivalent of one glucose molecule, two acetyl-CoA must be metabolized by the Krebs cycle. Two waste products, H2O and CO2, are created during this cycle.
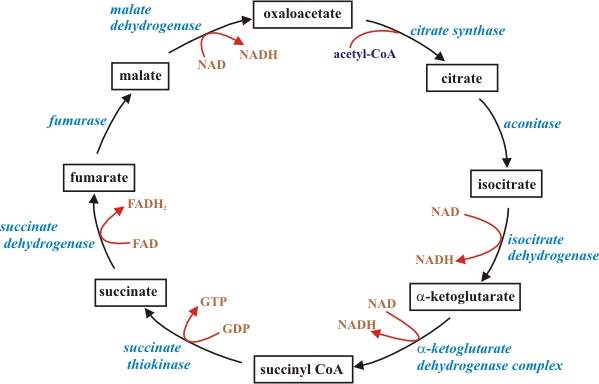
The citric acid cycle is an 8-step process involving 8 different enzymes. Throughout the entire cycle, acetyl-CoA changes into citrate, isocitrate, α-ketoglutarate, succinyl-CoA, succinate, fumarate, malate, and finally, oxaloacetate. The net energy gain from one cycle is 3 NADH, 1 FADH, and 1 ATP. Thus, the total amount of energy yield from one whole glucose molecule (2 pyruvate molecules) is 6 NADH, 2 FADH, and 2 ATP.
Oxidative Phosphorylation:
In eukaryotes, oxidative phosphorylation occurs in the mitochondrial cristae. It comprises the electron transport chain that establishes a proton gradient (chemiosmotic potential) across the inner membrane by oxidizing the NADH produced from the Krebs cycle. ATP is synthesised by the ATP synthase enzyme when the chemiosmotic gradient is used to drive the phosphorylation of ADP. The electrons are finally transferred to exogenous oxygen and, with the addition of two protons, water is formed.
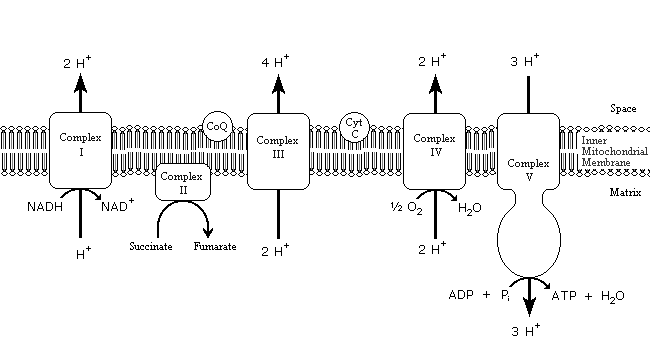
Electron Transport Chain (ETC):
During oxidative phosphorylation, electrons derived from NADH and FADH2 combine with O2, and the energy released from these oxidation/ reduction reactions is used to drive the synthesis of ATP from ADP.
Electrons from NADH enter the electron transport
chain in complex I, which consists of nearly 40 polypeptide chains. These
electrons are initially transferred from NADH to flavin mononucleotide and then,
through an iron-sulfur carrier, to coenzyme Ql. Coenzyme Q (also called
ubiquinone) is a small, lipid-soluble molecule that carries electrons from
complex I through the membrane to complex III, which consists of about ten
polypeptides. In complex III, electrons are transferred from cytochrome b
to cytochrome c![]() an
energy-yielding reaction. Cytochrome c, a peripheral membrane protein
bound to the outer face of the inner membrane, then carries electrons to complex
IV (cytochrome oxidase), where they are finally transferred to O2.
an
energy-yielding reaction. Cytochrome c, a peripheral membrane protein
bound to the outer face of the inner membrane, then carries electrons to complex
IV (cytochrome oxidase), where they are finally transferred to O2.
A distinct protein complex (II), which consists of four polypeptides, receives electrons from the citric acid cycle intermediate, succinate. These electrons are transferred to FADH2, rather than to NADH, and then to coenzyme Q. From coenzyme Q, electrons are transferred to complex III and then to complex IV as already described. In contrast to the transfer of electrons from NADH to coenzyme Q at complex I, the transfer of electrons from FADH2 to coenzyme Q is not associated with a significant decrease in free energy and, therefore, is not coupled to ATP synthesis. Consequently, the passage of electrons derived from FADH2 through the electron transport chain yields free energy only at complexes III and IV.
ATP synthesis:
The mechanism of coupling electron transport to ATP generation, chemiosmotic coupling, is a striking example of the relationship between structure and function in cell biology. The hypothesis of chemiosmotic coupling was first proposed in 1961 by Peter Mitchell, who suggested that ATP is generated by the use of energy stored in the form of proton gradients across biological membranes, rather than by direct chemical transfer of high-energy groups.
Electron transport through complexes I, III, and IV is coupled to the transport of protons out of the interior of the mitochondrion. Thus, the energy-yielding reactions of electron transport are coupled to the transfer of protons from the matrix to the intermembrane space, which establishes a proton gradient across the inner membrane. Complexes I and III each transfer four protons across the membrane per pair of electrons. n complex IV, two protons per pair of electrons are pumped across the membrane and another two protons per pair of electrons are combined with O2 to form H2O within the matrix. Thus, the equivalent of four protons per pair of electrons is transported out of the mitochondrial matrix at each of these three complexes.
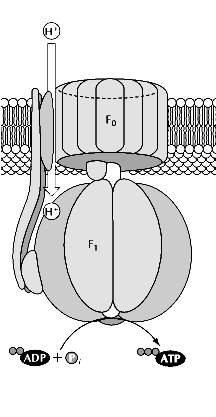
Because the phospholipid bilayer is impermeable to ions, protons are able to cross the membrane only through a protein channel. This restriction allows the energy in the electrochemical gradient to be harnessed and converted to ATP as a result of the action of the fifth complex involved in oxidative phosphorylation, complex V, or ATP synthase. ATP synthase is organized into two structurally distinct components, F0 and F1, which are linked by a slender stalk. The F0 portion spans the inner membrane and provides a channel through which protons are able to flow back from the intermembrane space to the matrix. The energetically favorable return of protons to the matrix is coupled to ATP synthesis by the F1 subunit, which catalyzes the synthesis of ATP from ADP and phosphate ions (Pi).
Transport across mitochondria:
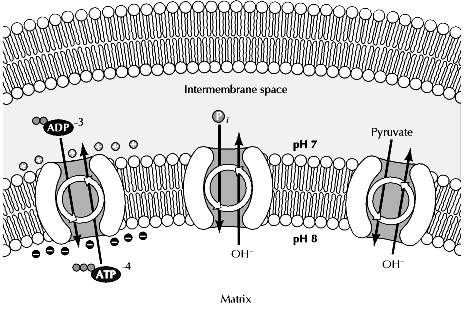
In addition to driving the synthesis of ATP, the potential energy stored in the electrochemical gradient drives the transport of small molecules into and out of mitochondria. For example, the ATP synthesized within mitochondria has to be exported to the cytosol, while ADP and Pi need to be imported from the cytosol for ATP synthesis to continue. The electrochemical gradient generated by proton pumping provides energy required for the transport of these molecules and other metabolites that need to be concentrated within mitochondria.
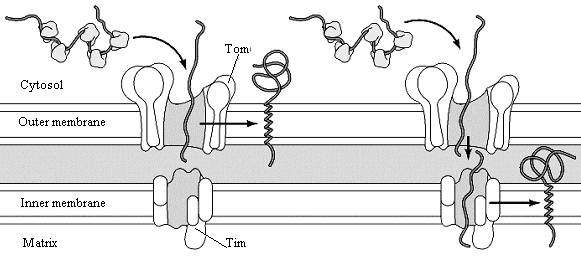
Most mitochondrial proteins are encoded by the nuclear genome. These proteins are translated on free ribosomes and imported into mitochondria as completed polypeptide chains. Positively charged presequences target proteins for import to the mitochondrial matrix. Phospholipids are carried to mitochondria from the endoplasmic reticulum by phospholipid transfer proteins.
THEORETICAL ENERGY YIELD:
It is now possible to calculate the total yield of ATP from the oxidation of glucose. The net gain from glycolysis is two molecules of ATP and two molecules of NADH. The conversion of pyruvate to acetyl CoA and its metabolism via the citric acid cycle yields two additional molecules of ATP, eight of NADH, and two of FADH2. Assuming that three molecules of ATP are derived from the oxidation of each NADH and two from each FADH2, the total yield is 38 molecules of ATP per molecule of glucose. However, this yield is lower in some cells because the two molecules of NADH generated by glycolysis in the cytosol are unable to enter mitochondria directly. Instead, their electrons must be transferred into the mitochondrion via a shuttle system. Depending on the system used, this transfer may result in these electrons entering the electron transport chain at the level of FADH2. In such cases, the two molecules of NADH derived from glycolysis give rise to two rather than three molecules of ATP, reducing the total yield to 36 rather than 38 ATPs per molecule of glucose.

Fermentation:
Fermentation is the process of deriving energy from the oxidation of organic compounds, such as carbohydrates, using an endogenous electron acceptor, which is usually an organic compound. Without oxygen, pyruvate is not metabolized by cellular respiration but undergoes a process of fermentation. The pyruvate is not transported into the mitochondrion, but remains in the cytoplasm, where it is converted to waste products that may be removed from the cell. This serves the purpose of oxidizing the hydrogen carriers so that they can perform glycolysis again and removing the excess pyruvate. This waste product varies depending on the organism. In skeletal muscles, the waste product is lactic acid. This type of fermentation is called lactic acid fermentation. In yeast, the waste products are ethanol and carbon dioxide. This type of fermentation is known as alcoholic or ethanol fermentation. The ATP generated in this process is made by substrate-level phosphorylation, which does not require oxygen
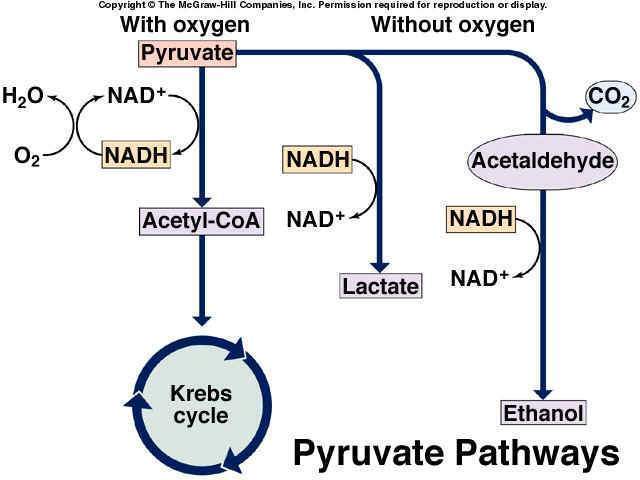
Anaerobic respiration:
Anaerobic respiration is used by some microorganisms, in which neither oxygen (aerobic respiration) nor pyruvate or pyruvate derivative (fermentation) is the final electron acceptor. Rather, an inorganic acceptor (for example, Sulfur) is used.